LECTURE 04
Body-Centred Cubic (BCC) Crystal Structure — Complete Analysis
Geometry, Derivations, Mechanical Properties & Engineering Applications By Dr. Rolly Verma | AdvanceMaterialsLab.com | March 2026 | B.Sc. / M.Sc. Materials Science & EngineeringTopic: BCC Crystal Structure — Full Structural Analysis | Level: Undergraduate / Postgraduate
Reading time: 40 minutes | Includes: Geometry, full derivations, void analysis, mechanical properties, applications, solved problems, MCQs
SEO Keywords: BCC crystal structure, body centred cubic, BCC atomic packing factor, BCC coordination number 8, body diagonal 4R a root 3, BCC vs FCC, iron crystal structure, refractory metals BCC
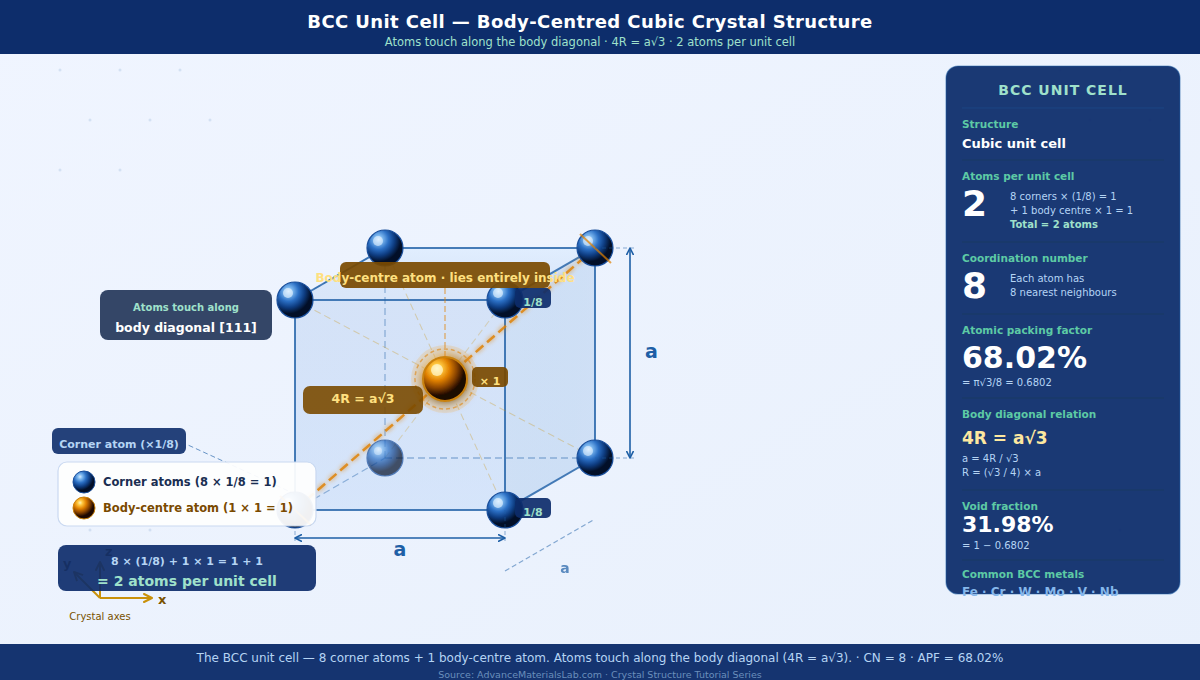
1. Introduction: What is the BCC Crystal Structure?
Welcome to this tutorial. If you have just begun your journey into materials science, one of the first and most important things you will encounter is the concept of a crystal structure — the organised, repeating arrangement of atoms defined by a lattice and basis inside a solid material. Understanding how atoms arrange themselves is the foundation for understanding virtually everything about a material's physical, mechanical, and electronic properties.
The Body-Centred Cubic (BCC) structure is one of the three most common crystal structures found in metallic elements, alongside Face-Centred Cubic (FCC) and Hexagonal Close-Packed (HCP). As the name suggests, BCC is a cubic arrangement with one special addition: an atom placed exactly at the geometric centre — the body centre — of the cube.
You can think of BCC as follows: imagine placing 8 identical billiard balls at the eight corners of a perfectly cubic wooden box, and then dropping one more ball directly into the centre of the box — pressing it down until it touches all 8 corner balls simultaneously. That mental picture captures the essence of the BCC unit cell perfectly. According to the authoritative textbook by Callister and Rethwisch — Materials Science and Engineering: An Introduction, BCC is one of the most widely studied crystal structures due to its prevalence in engineering metals.
- Iron (Fe) — the backbone of steel — adopts the BCC structure below 912°C. Understanding BCC is literally understanding the foundation of the global steel industry.
- Refractory BCC metals like tungsten (W), molybdenum (Mo), and chromium (Cr) are essential in high-temperature applications — jet engines, nuclear reactors, and cutting tools.
- BCC metals display a characteristic ductile-to-brittle transition at low temperatures — a phenomenon directly linked to their crystal structure and slip system geometry.
- The body diagonal touching direction in BCC determines its unique mechanical behaviour, distinct from FCC metals of comparable composition.
2. Cube Geometry and Lattice Parameters
The BCC structure belongs to the cubic crystal system — one of the seven crystal systems in crystallography. If you need a refresher on how crystal systems are defined through lattice geometry, our tutorial on Lattice and Basis in Crystallography covers this in full detail. The cubic system is further classified by the International Union of Crystallography (IUCr). In a cubic system, all three edge lengths are equal and all angles between edges are exactly 90°. This high degree of symmetry makes the BCC structure relatively straightforward to analyse mathematically.
The BCC unit cell is completely described by a single lattice parameter — the lattice constant a, which is the length of the cube edge. If you are new to the concept of unit cells and lattice parameters, we recommend reading our introductory tutorial on Unit Cell and Lattice Parameters before proceeding. All eight corners of the BCC cube are equivalent, and the body-centre position is the single unique interior site.
| Metal | Symbol | Lattice Parameter a (nm) | Atomic Radius R (pm) |
|---|---|---|---|
| Iron (alpha) | Fe | 0.2866 | 124 |
| Chromium | Cr | 0.2885 | 125 |
| Tungsten | W | 0.3165 | 137 |
| Molybdenum | Mo | 0.3147 | 136 |
| Vanadium | V | 0.3030 | 131 |
| Niobium | Nb | 0.3301 | 143 |
| Tantalum | Ta | 0.3306 | 143 |
Data source: NIST Crystal Structure Data
3. Atoms per Unit Cell in BCC
When we count atoms in any crystal unit cell, we must account for the fact that atoms sitting on the boundary of the cell are shared between neighbouring cells. This is the atom-sharing rule — a fundamental concept you should commit to memory.
In the BCC unit cell, there are two types of atomic positions:
- Corner atoms (8 total): Each corner atom sits at the intersection of 8 unit cells — it belongs equally to all 8. Therefore each corner atom contributes 1/8 of itself to our unit cell.
- Body-centre atom (1 total): This atom sits completely inside the unit cell. It belongs entirely to this one unit cell alone. It contributes a full 1.
Applying the atom-sharing rule:
N = (Number of corner atoms × 1/8) + (Number of body-centre atoms × 1)
N = (8 × 1/8) + (1 × 1)
N = 1 + 1 = 2 atoms per unit cell
Imagine 8 friends sitting at 8 corners of a square table, sharing a single small cake placed at each corner. Each person only gets 1/8 of that corner cake since it is shared equally. But the person sitting at the exact centre of the table has their own cake — they do not share with anyone. That is the BCC atom count: 8 people × 1/8 cake + 1 centre person × 1 full cake = 2 cakes total.
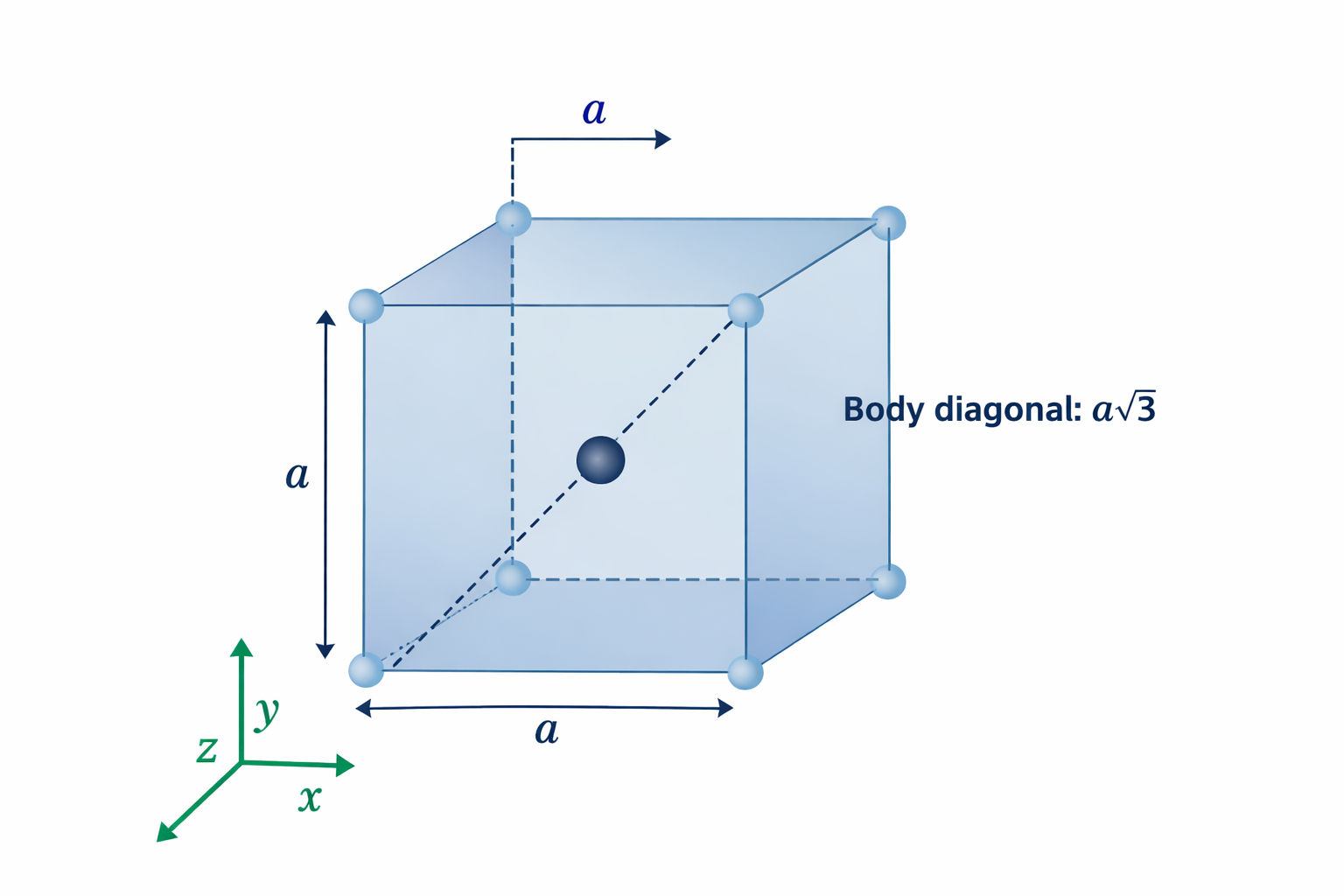
4. The Body Diagonal — The Touching Direction in BCC
In a crystal structure, atoms are modelled as hard spheres. They are assumed to be touching in at least one crystallographic direction — this is called the close-packed direction. Knowing which direction atoms touch is essential because it gives us the precise mathematical relationship between the lattice parameter a and the atomic radius R.
In BCC, atoms do not touch along the cube edge (as in Simple Cubic). They do not touch along the face diagonal (as in FCC). Instead, BCC atoms touch along the body diagonal — the line that cuts through the geometric centre of the cube from one corner to the diagonally opposite corner.
Imagine standing at one corner of a cube and looking towards the diagonally opposite corner — not along an edge, not across a face, but straight through the interior. That line is the body diagonal. In BCC, it passes through: corner atom → body-centre atom → corner atom at the far end. All three are touching.
4.1 Relationship Between Lattice Parameter a and Atomic Radius R
The body diagonal of a cube with edge length a has a length given by the three-dimensional Pythagorean theorem:
Body diagonal length = √(a² + a² + a²) = √(3a²) = a√3
Now, looking along the body diagonal, we see three atoms in contact: a corner atom (contributing one radius R from its centre to the cube corner), the body-centre atom (contributing its full diameter 2R), and the far corner atom (contributing one radius R). Therefore the body diagonal spans exactly 4R:
4R = a√3
Therefore:
a = 4R / √3 (lattice parameter in terms of R)
R = (√3 / 4) × a (atomic radius in terms of a)
R = a√3 / 4
You can verify this numerically for iron: a = 0.2866 nm, so R = a√3/4 = 0.2866 × 1.732 / 4 = 0.2866 × 0.433 = 0.1241 nm = 124.1 pm. The experimentally measured atomic radius of Fe is 124 pm — excellent agreement!
5. Coordination Number — Why CN = 8 in BCC
The coordination number (CN) is defined as the number of nearest-neighbour atoms that directly touch any given atom in the crystal. It is one of the most important structural parameters because it directly determines the number of bonds per atom and therefore the cohesive energy and many physical properties of the material.
To find the CN of a BCC atom, let us think about the body-centre atom specifically — it is the easiest one to visualise. The body-centre atom sits at the exact geometric centre of the cube. Its 8 nearest neighbours are the 8 corner atoms, and by the body diagonal relationship (4R = a√3), it is touching all 8 of them simultaneously.
CN (BCC) = 8
Nearest-neighbour distance = a√3/2
= 4R/2 = 2R (atoms touching — confirms our model)
In BCC, there are also 6 second-nearest neighbours — the 6 atoms at the face-centre positions of adjacent unit cells, at a distance of a from any given atom. These are only 15% further away than the nearest neighbours (distance a vs a√3/2 = 0.866a), which is unusually close. This near-degeneracy of the first and second coordination shells gives BCC metals their distinctive mechanical behaviour under stress.
Place one atom at the centre of a Rubik's cube. Each of the 8 corner sub-cubes touches the centre piece — that is 8 corners, each equally close. This is the BCC coordination: the body-centre atom is equidistant from all 8 corners, touching every single one. CN = 8.
6. Atomic Packing Factor — Complete Derivation
The Atomic Packing Factor (APF) — also called packing fraction or packing efficiency — is the fraction of the unit cell volume actually occupied by atoms. It tells us how efficiently atoms fill the available space.
APF = Volume occupied by atoms in unit cell / Total volume of unit cell
Step 1: Count the atoms
N = 2 atoms (as derived above)
Step 2: Volume of atoms in unit cell
V_atoms = N × (4/3)πR³ = 2 × (4/3)πR³ = (8/3)πR³
Step 3: Express the unit cell volume in terms of R
From 4R = a√3: a = 4R/√3
V_cell = a³ = (4R/√3)³ = (64R³) / (√3)³ = 64R³ / (3√3)
Step 4: Calculate APF
APF = V_atoms / V_cell
APF = [(8/3)πR³] / [64R³ / (3√3)]
APF = [(8/3)πR³] × [(3√3) / (64R³)]
APF = (8π√3) / (3 × 64/3)
APF = (8π√3) / 64
APF = π√3 / 8
APF (BCC) = π√3 / 8 = 3.14159 × 1.7321 / 8 = 5.4414 / 8 = 0.6802
This means 68.02% of the BCC unit cell volume is occupied by atoms, while the remaining 31.98% is empty interstitial space. Compare this to FCC and HCP (both 74.05%) and Simple Cubic (52.36%) — BCC falls in between, achieving moderate but not maximum packing efficiency.
In BCC, atoms along the cube edge are not touching — there is a gap between corner atoms along the edge direction. Only along the body diagonal are atoms in contact. This means a significant fraction of space along the face diagonals and cube edges remains empty. FCC achieves higher packing because it places additional atoms at face centres, filling these gaps efficiently. The trade-off: BCC's open structure allows faster diffusion and gives distinct slip behaviour.
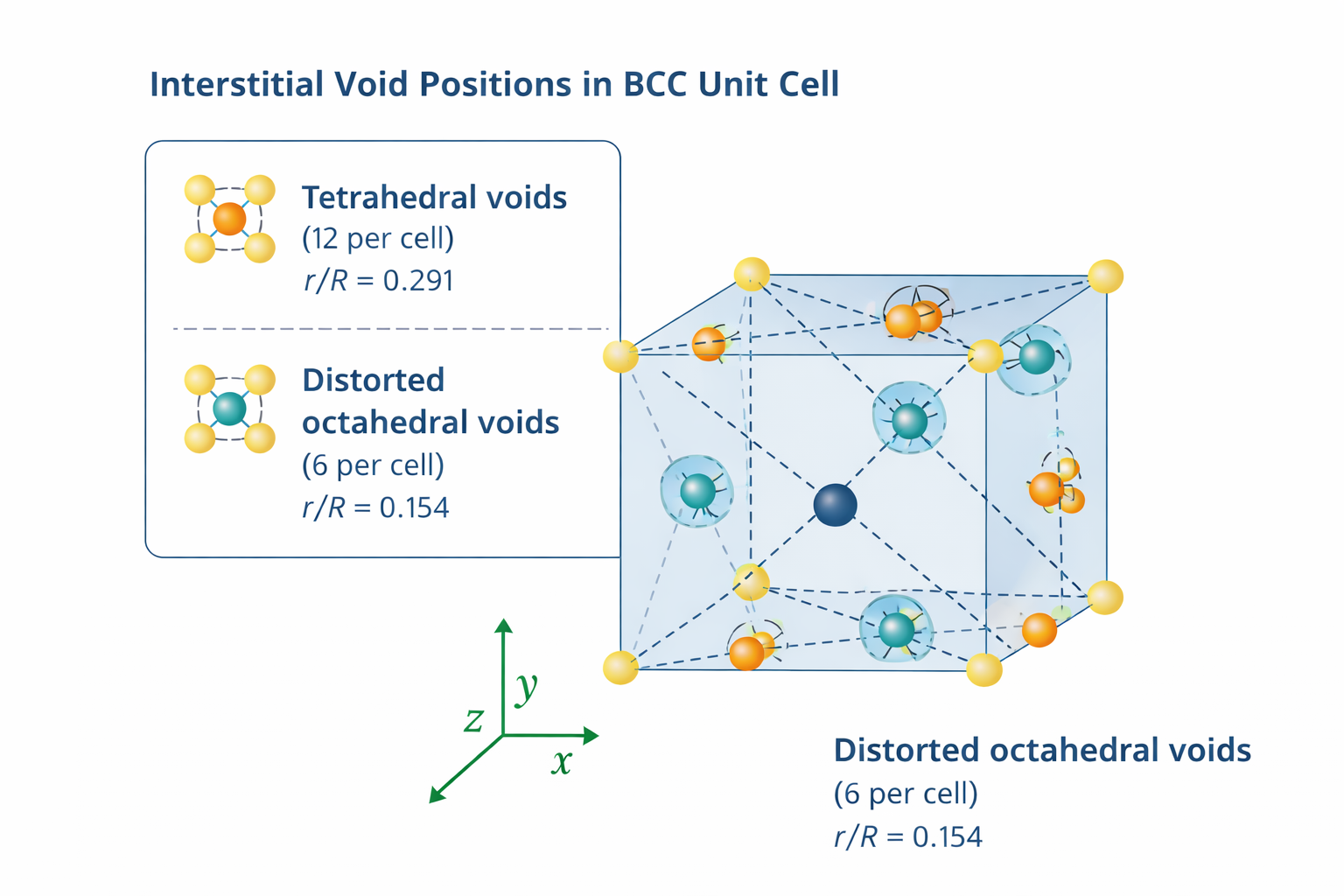
7. Interstitial Voids in BCC Structure
The empty spaces between atoms in a crystal are called interstitial voids or interstitial sites. These are critically important in materials science because small atoms (like carbon, nitrogen, hydrogen, and boron) can fit into these spaces, dramatically altering the material's properties. Steel — arguably the most important engineering material in human history — owes its remarkable properties to carbon atoms occupying interstitial voids in the iron crystal structure.
BCC has two types of interstitial voids:
7.1 Tetrahedral Voids
A tetrahedral void is a void space surrounded by 4 atoms arranged at the corners of a regular tetrahedron. In BCC, tetrahedral voids are relatively large and are the preferred sites for interstitial atoms at low concentrations.
Number of tetrahedral voids per BCC unit cell = 12
Radius of tetrahedral void (r_t):
r_t / R = 0.291 → r_t = 0.291 R
7.2 Octahedral Voids
An octahedral void is surrounded by 6 atoms at the corners of a regular octahedron. Surprisingly, in BCC the octahedral voids are distorted — they are not regular octahedra. This distortion means the octahedral voids in BCC are actually smaller than the tetrahedral voids, which is the opposite of what occurs in FCC.
Number of octahedral voids per BCC unit cell = 6
Radius of octahedral void (r_o):
r_o / R = 0.154 → r_o = 0.154 R
Carbon atoms (radius ≈ 77 pm) occupy the distorted octahedral voids in BCC alpha-iron (not tetrahedral, as is often assumed). The octahedral void radius in BCC Fe is about 0.154 × 124 = 19 pm — far smaller than the carbon atom radius of 77 pm. This severe mismatch means carbon creates enormous lattice strain when it dissolves in BCC iron, which is why the solubility of carbon in alpha-iron is only 0.022 wt% at 727°C. In contrast, carbon in FCC gamma-iron (austenite) can dissolve up to 2.14 wt% due to the larger, more regular interstitial sites.
This difference in interstitial solubility between BCC alpha-iron and FCC gamma-iron is the physical basis for all of steel metallurgy and heat treatment — as discussed in detail by DoITPoMS (University of Cambridge) — Iron-Carbon Phase Diagram.
8. Theoretical Density of BCC Metals
The theoretical density of a crystalline material is calculated directly from its crystal structure data. For BCC materials, the formula is:
ρ = (n × A) / (V_c × N_A)
Where:
n = atoms per unit cell = 2 (BCC)
A = atomic mass (g/mol) — from periodic table
V_c = a³ = (4R/√3)³ (in cm³)
N_A = Avogadro's number = 6.022 × 10²³ atoms/mol
The lattice parameter a is given in nm or Å in data tables. Convert to cm before calculating V_c:
1 nm = 10⁻⁷ cm | 1 Å = 10⁻⁸ cm
Example for iron: a = 0.2866 nm = 2.866 × 10⁻⁸ cm → V_c = (2.866 × 10⁻⁸)³ = 23.55 × 10⁻²⁴ cm³
9. Mechanical Properties of BCC Metals
The crystal structure of BCC metals directly determines their mechanical behaviour in ways that distinguish them sharply from FCC metals. Understanding these connections is central to materials engineering and is covered extensively in resources like the ASM International Metals Handbook.
9.1 Slip Systems in BCC
Plastic deformation in metals occurs primarily by slip — the sliding of one plane of atoms over another along a specific crystallographic direction. The combination of a slip plane and a slip direction is called a slip system. The number and arrangement of slip systems in a crystal directly determines its ductility and formability.
In BCC, the close-packed direction (the direction along which atoms touch) is <111> — the body diagonal direction. The primary slip planes are {110}, {112}, and {123}.
BCC primary slip systems:
{110}⟨111⟩ — 6 planes × 2 directions = 12 slip systems
{112}⟨111⟩ — 12 planes × 1 direction = 12 slip systems
{123}⟨111⟩ — 24 planes × 1 direction = 24 slip systems
Total: up to 48 slip systems
Although BCC has more slip systems than FCC (12), its slip planes are not close-packed — they have lower atomic density than the {111} planes in FCC. This creates higher resistance to dislocation motion (higher Peierls stress), which is why BCC metals tend to be stronger but less ductile than FCC metals of similar composition.
9.2 Ductile-to-Brittle Transition Temperature (DBTT)
One of the most practically important properties of BCC metals is their ductile-to-brittle transition temperature (DBTT). Above the DBTT, BCC metals are ductile and tough. Below it, they become brittle and fracture suddenly without significant plastic deformation.
FCC metals do NOT exhibit a DBTT — they remain ductile down to absolute zero. HCP metals show a partial transition depending on their c/a ratio and available slip systems. The DBTT in BCC metals arises because the Peierls stress for dislocation glide in BCC increases steeply at low temperatures, eventually exceeding the cleavage fracture stress.
During World War II, several US Liberty Ships — made from low-carbon BCC steel — fractured catastrophically in cold North Atlantic waters. The cold temperatures pushed the steel below its DBTT, causing sudden brittle fracture instead of ductile yielding. This led to the development of fracture mechanics as a formal engineering discipline, and is now a classic example taught in every materials engineering curriculum.
9.3 High Melting Points of BCC Refractory Metals
Many refractory metals (metals with very high melting points) adopt the BCC structure. This is because the <111> bonding direction in BCC involves a high degree of orbital overlap between adjacent atoms, creating particularly strong metallic bonds.
| BCC Metal | Melting Point (°C) | Primary Application | Key Property |
|---|---|---|---|
| Tungsten (W) | 3422 | Light bulb filaments, X-ray targets | Highest melting point of all metals |
| Rhenium (Re) | 3186 | Jet engine superalloys | Excellent creep resistance |
| Molybdenum (Mo) | 2623 | Steel alloying, electronics | Low thermal expansion |
| Niobium (Nb) | 2477 | Superconducting magnets | Superconducting below 9.3 K |
| Chromium (Cr) | 1907 | Stainless steel, hard coatings | Excellent oxidation resistance |
| Iron (Fe, α) | 1538 | Structural steel, magnets | Ferromagnetic, ubiquitous |
| Vanadium (V) | 1910 | High-strength steels, nuclear | Low neutron cross-section |
10. Common BCC Metals and Their Engineering Applications
10.1 Iron — The Most Important BCC Metal
Iron exhibits a fascinating structural transformation called allotropy — it exists in different crystal structures at different temperatures. At room temperature and up to 912°C, iron is BCC (called alpha-iron or ferrite). Between 912°C and 1394°C, it transforms to FCC (called gamma-iron or austenite). Above 1394°C it reverts to BCC (called delta-iron) until it melts at 1538°C.
Iron allotropic transformations:
α-Fe (BCC) → γ-Fe (FCC) → δ-Fe (BCC) → Liquid
912°C 1394°C 1538°C
The BCC ↔ FCC transformation in iron is the physical basis for all steel heat treatment processes — annealing, quenching, tempering, and case hardening. Without this structural transformation, the entire science of steel metallurgy would not exist. More details on the iron-carbon system are available from the ScienceDirect resource on the Iron-Carbon Phase Diagram.
10.2 Chromium — The Corrosion-Resistant BCC Metal
Chromium (BCC, a = 0.2885 nm) is the essential alloying element in stainless steel. When at least 10.5 wt% Cr is added to iron, a self-healing passive oxide layer (Cr₂O₃) forms on the surface, giving the alloy its characteristic corrosion resistance. Chromium's BCC structure is compatible with alpha-iron's BCC lattice, allowing complete solid solubility in the ferritic (BCC) phase.
10.3 Tungsten — The Refractory BCC Metal
Tungsten has the highest melting point of any element (3422°C) and maintains BCC structure at all temperatures up to melting. It is used in light bulb filaments, X-ray tube anodes, TIG welding electrodes, and as a shielding material in radiation environments. Its exceptionally high density (19.3 g/cm³) also makes it valuable as a kinetic energy penetrator in armour-piercing projectiles.

11. BCC vs FCC vs HCP — Master Comparison Table
Now that you have a thorough understanding of BCC, let us place it in context by comparing it systematically with the other two major metallic crystal structures. This comparison table is your definitive quick reference for examinations and research. For a deeper treatment, read our dedicated tutorials on FCC Crystal Structure and HCP Crystal Structure on AdvanceMaterialsLab.com.
| Property | BCC | FCC | HCP |
|---|---|---|---|
| Atoms per unit cell | 2 | 4 | 6 |
| Coordination number | 8 | 12 | 12 |
| Touching direction | Body diagonal <111> | Face diagonal <110> | Basal plane <1120> |
| a vs R | a = 4R/√3 | a = 2√2 R | a = 2R |
| APF (exact) | π√3/8 | π/(3√2) | π/(3√2) |
| APF (%) | 68.02% | 74.05% | 74.05% |
| Void fraction | 31.98% | 25.95% | 25.95% |
| Close-packed? | No | Yes | Yes |
| Primary slip plane | {110}, {112}, {123} | {111} | {0001} basal |
| Slip systems | Up to 48 | 12 | 3 (basal) |
| Typical ductility | Moderate | High | Low–Moderate |
| DBTT | Yes (characteristic) | No | Partial |
| Typical melting points | High (refractory) | Moderate | Variable |
| Example metals | Fe, Cr, W, Mo, V, Nb | Al, Cu, Au, Ni, Ag | Mg, Ti, Zn, Co |
SC (52%) < BCC (68%) < FCC = HCP (74%)
BCC occupies the middle ground — more efficiently packed than Simple Cubic, but less so than the close-packed structures. Its moderate packing creates a 32% void fraction that is critically important for interstitial diffusion in steels and other engineering alloys.
12. Solved Problems — Step by Step
For BCC, atoms touch along the body diagonal: 4R = a√3
R = a√3 / 4 = (0.2885 × 1.7321) / 4
R = 0.4997 / 4 = 0.1249 nm = 124.9 pm ✔
Experimentally measured atomic radius of Cr = 125 pm — perfect agreement!
n = 2 (BCC)
a = 0.2866 nm = 2.866 × 10⁻⁸ cm
V_c = a³ = (2.866 × 10⁻⁸)³ = 23.55 × 10⁻²⁴ cm³
ρ = (n × A) / (V_c × N_A)
ρ = (2 × 55.85) / (23.55 × 10⁻²⁴ × 6.022 × 10²³)
ρ = 111.70 / (23.55 × 10⁻²⁴ × 6.022 × 10²³)
ρ = 111.70 / 14.18 = 7.87 g/cm³ ✔
Experimentally measured density of iron = 7.87 g/cm³ — exact match!
Step 1: n = 2 atoms (BCC)
Step 2: V_atoms = 2 × (4/3)πR³ = (8/3)πR³
Step 3: a = 4R/√3 → a³ = (4R/√3)³ = 64R³/(3√3)
Step 4: APF = (8/3)πR³ / [64R³/(3√3)]
= (8π/3) × (3√3/64)
= 8π√3 / 64
= π√3 / 8
= 3.14159 × 1.7321 / 8
= 0.6802 = 68.02% ✔
Side of crystal = 1 mm = 1 × 10⁶ nm
Lattice parameter a = 0.2866 nm
Number of unit cells along one edge = 10⁶ / 0.2866 = 3.489 × 10⁶
Total unit cells = (3.489 × 10⁶)³ = 4.24 × 10¹⁹ unit cells
Total atoms = 4.24 × 10¹⁹ × 2 = 8.48 × 10¹⁹ atoms ✔
This illustrates the enormous number of atoms even in a tiny crystal — a 1 mm cube of iron contains approximately 85 million trillion atoms.
For BCC octahedral void: r_void = 0.154 × R
r_void = 0.154 × 124 pm = 19.1 pm
Maximum atom diameter = 2 × r_void = 38.2 pm
Carbon atomic radius = 77 pm >> 38.2 pm
→ Carbon DOES distort the BCC lattice significantly
→ This limits carbon solubility in alpha-iron to only 0.022 wt% ✔
13. Practice Multiple Choice Questions
- (a) Cube edge <100>
- (b) Body diagonal <111> ✔ — 4R = a√3
- (c) Face diagonal <110>
- (d) They do not touch in any direction
- (a) π/6 = 52.36%
- (b) π√3/8 = 68.02% ✔
- (c) π/(3√2) = 74.05%
- (d) π/4 = 78.54%
- (a) 1
- (b) 2 ✔ — 8×(1/8) + 1×1 = 2
- (c) 4
- (d) 6
- (a) a/2
- (b) a√2/4
- (c) a√3/4 ✔ — from 4R = a√3
- (d) a√2/2
- (a) The 6 face-centre atoms of adjacent unit cells
- (b) The 8 corner atoms of the same unit cell ✔
- (c) The 4 atoms in the same close-packed plane
- (d) The body-centre atoms of the 8 adjacent unit cells
- (a) Carbon and iron have very different atomic masses
- (b) The octahedral interstitial voids in BCC iron are much smaller than in FCC iron ✔
- (c) BCC iron has fewer unit cells per volume
- (d) Carbon prefers hexagonal crystal structures
- (a) High electrical conductivity
- (b) Ability to undergo plastic deformation
- (c) Ductile-to-brittle transition temperature (DBTT) ✔
- (d) Formation of interstitial solid solutions
- (a) APF decreases from 74% to 68%
- (b) APF increases from 68% to 74% — atoms pack more efficiently ✔
- (c) APF remains unchanged
- (d) APF drops to 52% during the transformation
- (a) It means BCC metals are less dense than FCC metals of the same element
- (b) The larger void fraction enables faster interstitial diffusion in BCC vs FCC ✔
- (c) It means BCC cannot form alloys with other metals
- (d) The void fraction has no practical significance
- (a) 1
- (b) 2 ✔ — BCC always has 2 atoms per unit cell
- (c) 4
- (d) 6
14. Key Takeaways
- BCC DEFINITION: A cubic unit cell with atoms at the 8 corners and 1 atom at the exact geometric centre — the body centre.
- ATOMS/CELL: N = 8×(1/8) + 1×1 = 2 atoms per unit cell.
- TOUCHING DIRECTION: Atoms touch along the body diagonal [111]. Body diagonal length = a√3 = 4R.
- a vs R RELATION: a = 4R/√3 = 2.309R | R = a√3/4 = 0.433a.
- COORDINATION NUMBER: CN = 8 — each atom touches 8 nearest neighbours along the 8 body diagonals of adjacent cells.
- APF: 68.02% = π√3/8. Higher than SC (52%) but lower than FCC and HCP (74%). BCC is not close-packed.
- VOID FRACTION: 31.98% — the largest of the three common cubic/hexagonal structures. Enables faster interstitial diffusion.
- INTERSTITIAL VOIDS: 12 tetrahedral (r/R = 0.291) + 6 distorted octahedral (r/R = 0.154) per unit cell. Octahedral voids in BCC are smaller than tetrahedral — opposite of FCC.
- MECHANICAL BEHAVIOUR: BCC metals show moderate ductility, high strength at room temperature, and a characteristic ductile-to-brittle transition temperature (DBTT) absent in FCC metals.
- KEY EXAMPLES: Fe (steel, magnets), Cr (stainless steel), W (high-temperature), Mo (electronics), V and Nb (high-strength alloys, superconductors).
- IRON ALLOTROPY: Iron is BCC (α-Fe) below 912°C and above 1394°C (δ-Fe). This BCC↔FCC transformation is the physical basis of all steel heat treatment.
- DENSITY FORMULA: ρ = (2 × A) / (a³ × N_A). Always convert a to cm before calculating.
Glossary
- Body-Centred Cubic (BCC)
- A crystal structure with atoms at the 8 corners of a cube and 1 atom at the geometric centre. Two atoms per unit cell.
- Lattice parameter (a)
- The edge length of the cubic unit cell. For BCC: a = 4R/√3. Typically measured in nm or Å.
- Body diagonal
- The line connecting two diagonally opposite corners of a cube, passing through the body centre. Length = a√3. The touching direction in BCC.
- Coordination number (CN)
- The number of nearest-neighbour atoms that directly touch a given atom. In BCC, CN = 8.
- Atomic Packing Factor (APF)
- The fraction of unit cell volume occupied by atoms. BCC APF = π√3/8 = 0.6802.
- Interstitial void
- Empty spaces between atoms in a crystal that can accommodate smaller atoms. BCC has tetrahedral (r/R = 0.291) and distorted octahedral (r/R = 0.154) voids.
- Allotropy
- The ability of an element to exist in more than one crystal structure. Iron undergoes BCC ↔ FCC allotropic transformation at 912°C.
- Slip system
- The combination of a crystallographic slip plane and slip direction along which plastic deformation (dislocation glide) occurs.
- DBTT
- Ductile-to-Brittle Transition Temperature — the temperature below which a BCC metal changes from ductile to brittle fracture behaviour.
- Refractory metal
- A metal with an exceptionally high melting point (generally above 2000°C). Most refractory metals (W, Mo, Nb, Ta, Re) adopt the BCC structure.
- Peierls stress
- The minimum stress needed to move a dislocation through a crystal. Higher in BCC than FCC, explaining BCC's greater strength and DBTT behaviour.
References and Further Reading
All references below are peer-reviewed journals, internationally recognised textbooks, or authoritative academic databases. They are provided in IEEE citation style for academic authenticity.
-
W. D. Callister Jr. and D. G. Rethwisch, Materials Science and Engineering: An Introduction, 10th ed. Hoboken, NJ, USA: John Wiley & Sons, 2018, pp. 40–78.
[Publisher: Wiley]
Primary textbook reference for BCC unit cell, APF derivation, lattice parameters, and theoretical density. -
J. F. Shackelford, Introduction to Materials Science for Engineers, 8th ed. Upper Saddle River, NJ, USA: Pearson Prentice Hall, 2015, pp. 52–95.
[Publisher: Pearson]
Secondary textbook reference covering crystal geometry, coordination numbers, and packing efficiency in cubic structures. -
C. Kittel, Introduction to Solid State Physics, 8th ed. Hoboken, NJ, USA: John Wiley & Sons, 2005, ch. 1, pp. 1–26.
[Publisher: Wiley]
Classic solid-state physics reference for Bravais lattices, BCC crystal structure, and lattice parameter relationships. -
International Union of Crystallography (IUCr), "Teaching Pamphlets: Crystal Symmetry and Space Groups," IUCr Education Pamphlets, no. 9. Chester, UK: IUCr, 2002.
[iucr.org — full text]
Authoritative reference for the seven crystal systems, space groups, and cubic crystal symmetry classifications. -
A. Jain et al., "Commentary: The Materials Project: A materials genome approach to accelerating materials innovation," APL Mater., vol. 1, no. 1, Art. no. 011002, Jul. 2013, doi: 10.1063/1.4812323.
[BCC Fe crystal data — Materials Project mp-13]
Source for verified BCC iron crystal structure data: space group Im-3m (229), lattice parameter, and bonding geometry. -
W. Pepperhoff and M. Acet, "The Structure of Iron," in Constitution and Magnetism of Iron and Its Alloys, ser. Engineering Materials. Berlin, Germany: Springer, 2001, ch. 1, pp. 1–13, doi: 10.1007/978-3-662-04345-5_1.
[SpringerLink]
Definitive Springer reference covering BCC alpha-iron, FCC gamma-iron, and delta-iron allotropic transformations with precise transition temperatures. -
University of Cambridge, DoITPoMS, "Introduction to Mechanical Properties of Materials — Ductile-Brittle Transition Temperature," Teaching and Learning Packages, Cambridge, UK: University of Cambridge, 2023.
[doitpoms.ac.uk]
Peer-reviewed Cambridge academic resource on DBTT in BCC metals, Peierls stress, and the Liberty Ship fracture case study. -
H. Luo, W. Guo, and F. Liu, "Ductile–Brittle Transition Mechanism and Dilute Solution Softening Effect of Body-Centered Cubic Metals," Metals, vol. 15, no. 7, Art. no. 743, Jun. 2025, doi: 10.3390/met15070743.
[MDPI Open Access]
Recent peer-reviewed review article (2025) covering DBTT mechanism in BCC metals via screw dislocation mobility, kink-pair nucleation, and Peierls potential. -
S. Takaki, K. Kawasaki, and Y. Kimura, "Mechanical properties of ultra fine grained steels," J. Mater. Process. Technol., vol. 117, no. 3, pp. 359–363, Nov. 2001, doi: 10.1016/S0924-0136(01)00783-4.
[ScienceDirect — DBTT overview]
Journal reference on ductile-to-brittle transition behaviour in BCC ferritic steels with grain refinement effects. -
C. S. Barrett and T. B. Massalski, Structure of Metals: Crystallographic Methods, Principles and Data, 3rd ed. Oxford, UK: Pergamon Press, 1980, pp. 1–68.
Classic crystallography textbook reference for BCC interstitial void geometry, tetrahedral and octahedral hole radius ratios, and hard sphere model derivations. -
University of Cambridge, DoITPoMS, "Lattice Energies, Iron-Carbon System," Teaching and Learning Packages. Cambridge, UK: University of Cambridge, 2023.
[doitpoms.ac.uk — Iron-Carbon Phase Diagram]
Authoritative academic resource on carbon solubility differences between BCC alpha-iron (0.022 wt%) and FCC gamma-iron (2.14 wt%) and steel metallurgy. -
ASM International, ASM Handbook, Volume 2: Properties and Selection — Nonferrous Alloys and Special-Purpose Materials. Materials Park, OH, USA: ASM International, 1990.
[asminternational.org]
Industry-standard reference for mechanical and physical properties of BCC refractory metals: W, Mo, Cr, Nb, Ta, and V. -
R. Verma and S. K. Rout, "Frequency-dependent ferro–antiferro phase transition and internal bias field influenced piezoelectric response of donor and acceptor doped bismuth sodium titanate ceramics," J. Appl. Phys., vol. 126, no. 9, Art. no. 094103, Sep. 2019, doi: 10.1063/1.5111505.
[AIP Publishing — DOI: 10.1063/1.5111505]
Author's peer-reviewed research on crystal structure analysis in perovskite ferroelectric materials (BNT ceramics), demonstrating expertise in structural characterisation methods covered in this tutorial. -
R. Verma and S. K. Rout, "Influence of annealing temperature on the existence of polar domain in uniaxially stretched polyvinylidene-co-hexafluoropropylene for energy harvesting applications," J. Appl. Phys., vol. 128, no. 23, Art. no. 234104, Dec. 2020, doi: 10.1063/5.0022463.
[AIP Publishing — DOI: 10.1063/5.0022463]
Author's peer-reviewed research on crystal structure and phase analysis in polymer ferroelectric systems.
This tutorial is part of the Crystal Structure Master Hub at AdvanceMaterialsLab.com
Next in series: FCC Crystal Structure | Miller Indices | XRD & Bragg's Law
© 2026 AdvanceMaterialsLab.com — Dr. Rolly Verma | All rights reserved
Dr. Rolly Verma
If you notice any inaccuracies or have constructive suggestions to improve the content, I warmly welcome your feedback. It helps maintain the quality and clarity of this educational resource. You can reach me at: advancematerialslab27@gmail.com
