LECTURE 04
Face-Centred Cubic (FCC) Crystal Structure
Unit Cell Geometry, Coordination Number, APF Derivation, Slip Systems, Interstitial Sites & Engineering Applications By Dr. Rolly Verma | AdvanceMaterialsLab.com | March 2026 | B.Sc. / M.Sc. Materials Science & EngineeringSeries: Crystal Structure Hub | Level: Undergraduate / Postgraduate | Prerequisites: Crystal Structure Intro, Lattice & Basis, Unit Cell & Lattice Parameters
Reading time: 40 minutes | Includes: Full geometry, atom counting, APF derivation, CN = 12 proof, slip systems, octahedral/tetrahedral voids, ABCABC stacking, real metals data, worked numericals, MCQs, key takeaways
SEO Keywords: FCC crystal structure, face centred cubic, FCC coordination number 12, FCC atomic packing factor 74 percent, FCC slip systems 12, octahedral tetrahedral interstitial sites FCC, face centred cubic unit cell 4 atoms
1. Introduction — Why FCC Is One of the Most Important Crystal Structures
Welcome to this complete analysis of the Face-Centred Cubic (FCC) crystal structure. In the previous lectures of this series, we built the conceptual and mathematical framework of crystallography: what crystal structure means, how any crystal is described by a lattice plus basis, and how the six lattice parameters quantify the geometry of the unit cell. Now we apply this framework to one of the three most important metallic crystal structures — the Face-Centred Cubic (FCC) structure.
FCC is significant for three reasons that make it impossible to study materials science without a thorough understanding of it:
- It governs the most ductile and electrically conductive engineering metals: Copper, aluminium, gold, silver, and nickel — which between them account for the majority of the world's electrical wiring, electronic devices, aerospace structures, and jewellery — all crystallise in the FCC structure. Their remarkable ductility, formability, and conductivity are direct consequences of the FCC geometry.
- It achieves the maximum possible packing efficiency: FCC shares the theoretical maximum packing fraction of 74.05% with the HCP structure — meaning no crystal arrangement of equal spheres can pack atoms more efficiently. Understanding why FCC achieves this limit, and what it means physically, is a fundamental lesson in geometric optimisation.
- It possesses 12 slip systems — more than any other common crystal structure — which explains why FCC metals are the most ductile of all metallic crystal structures. This directly underpins the engineering of every flexible metal conductor, every drawn wire, and every rolled sheet in modern technology.
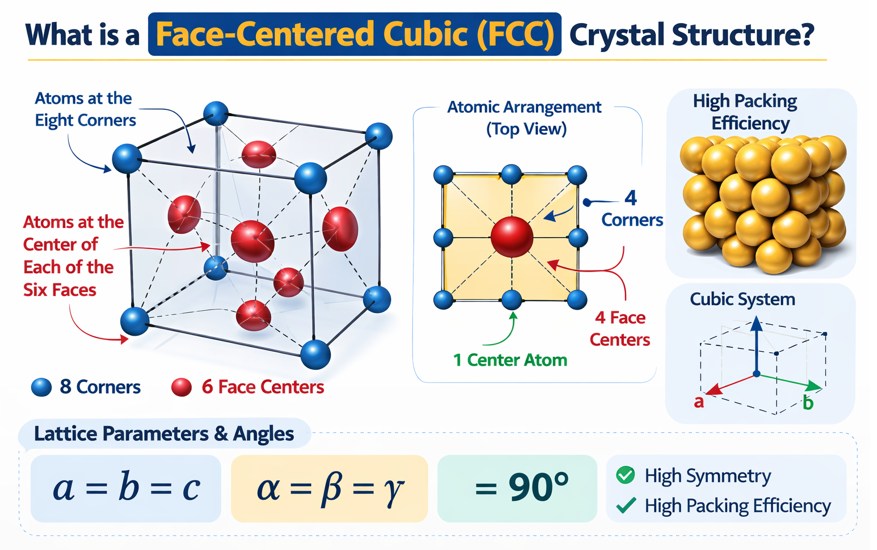
2. Visualising the FCC Unit Cell
The FCC unit cell belongs to the cubic crystal system, which means all three edge lengths are equal (a = b = c) and all interaxial angles are 90° (α = β = γ = 90°). So the unit cell is a perfect cube. The distinctive feature of FCC — what separates it from Simple Cubic and BCC — is the placement of atoms.
Imagine looking at the face of a cube from directly above. You see a square. In Simple Cubic, there is only an atom at each corner of that square. In BCC, there is additionally an atom at the very centre of the square's interior. In FCC, there is an atom at the centre of each face of the cube — like the central button on a cushion, sitting exactly halfway between the four corner atoms on each face. Repeat this for all six faces, and you have the FCC unit cell.
2.1 Atomic Positions in the FCC Unit Cell
In fractional coordinates (each coordinate expressed as a fraction of the lattice parameter a), the atomic positions in the FCC unit cell are:
Corner atoms (8 positions — shared with 7 other unit cells):
(0,0,0), (1,0,0), (0,1,0), (0,0,1),
(1,1,0), (1,0,1), (0,1,1), (1,1,1)
Face-centre atoms (6 positions — shared with 1 adjacent unit cell):
(½,½,0), (½,0,½), (0,½,½),
(½,½,1), (½,1,½), (1,½,½)
2.2 FCC as Lattice Plus Basis
Using the framework from Lecture 02, the FCC crystal structure is described as:
The FCC lattice is one of the 14 Bravais lattices. For elemental FCC metals (Cu, Al, Au, Ag, Ni), the basis is a single atom at (0,0,0). The FCC lattice itself already encodes the face-centre positions through its primitive vectors. This distinguishes elemental FCC metals (1-atom basis) from the diamond cubic structure (2-atom basis on FCC lattice) or NaCl (2-atom basis on FCC lattice with different species).
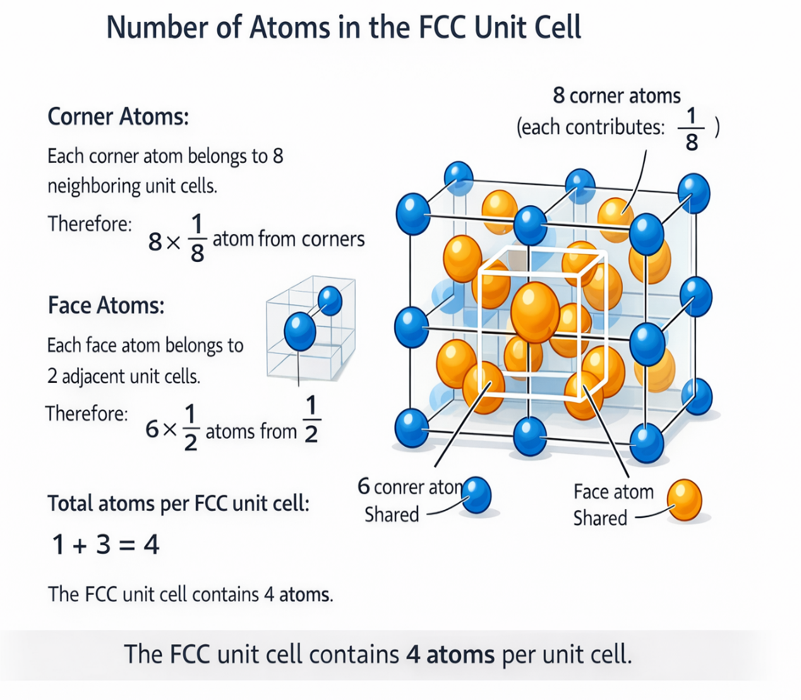
3. Atoms per Unit Cell — Full Derivation
Using the atom-sharing rule from crystallography:
Step 1: Corner atoms
Each corner of the cube is shared equally among 8 adjacent unit cells. Therefore each corner atom contributes 1/8 to our unit cell. With 8 corners:
8 corners × (1/8) = 1 atom
Step 2: Face-centre atoms
Each face-centre atom sits on a face that is shared between exactly 2 adjacent unit cells. Therefore each face atom contributes 1/2. With 6 faces:
6 face-centres × (1/2) = 3 atoms
Step 3: Total atoms per FCC unit cell
N_FCC = 1 (from corners) + 3 (from face-centres) = 4 atoms
This result — 4 atoms per conventional FCC unit cell — is one of the most important numbers in materials science. It means FCC has 4 times as many atoms as Simple Cubic (1 atom) and twice as many as BCC (2 atoms) in their respective conventional unit cells.
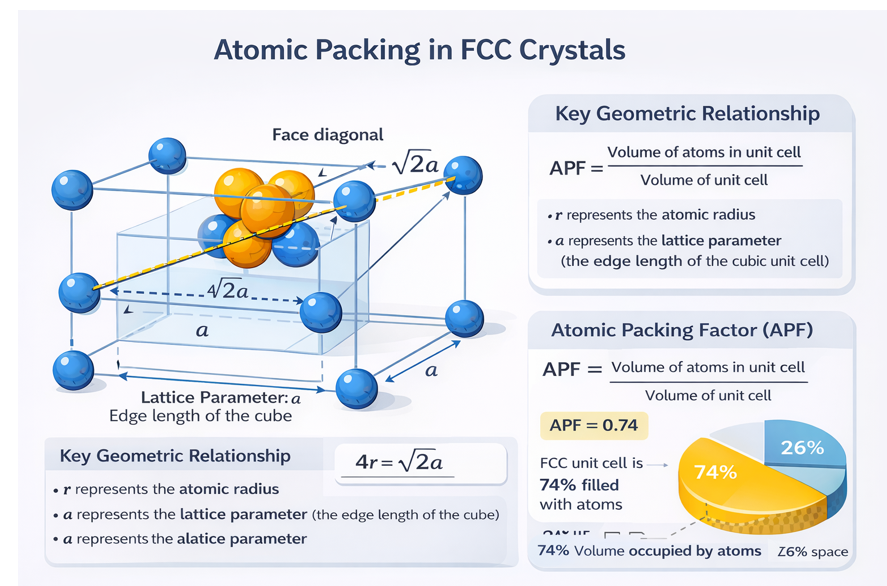
4. The Face Diagonal — The Touching Direction in FCC
In the hard sphere model, we assume atoms are rigid spheres that touch in at least one direction. In FCC, atoms do not touch along the cube edge (there is a gap between corner atoms) and do not touch along the body diagonal. Instead, FCC atoms touch along the face diagonal — the line connecting two opposite corners of any cube face, passing through the face-centre atom.
Look at any one face of the FCC cube. You see four corner atoms and one face-centre atom. Draw a diagonal from one corner to the diagonally opposite corner — it passes directly through the face-centre atom. Along this diagonal: corner atom (radius R) → face-centre atom (diameter 2R) → corner atom (radius R). The diagonal therefore spans 4R. Since the face diagonal of a square of side a equals a√2, we have 4R = a√2.
4.1 Relationship Between Lattice Parameter a and Atomic Radius R
The face diagonal of a square face of side a has length a√2. Counting the atomic contact sequence along this diagonal:
4R = a√2
Therefore:
a = 4R / √2 = 2√2 R (lattice parameter in terms of R)
R = a / (2√2) = a√2 / 4 = 0.3536 a (atomic radius in terms of a)
Copper: a = 0.3615 nm. From 4R = a√2: R = a√2/4 = 0.3615 × 1.4142 / 4 = 0.5115 / 4 = 0.1279 nm = 127.9 pm. The tabulated atomic radius of copper is 128 pm — perfect agreement.
5. Coordination Number — Why CN = 12 in FCC
The coordination number (CN) is the number of atoms directly touching any given atom in the crystal. In FCC, every atom has exactly 12 nearest neighbours — the maximum possible for a single-component crystal structure of equal-sized spheres. This maximum coordination is why FCC and HCP are both called close-packed structures.
5.1 How the 12 Nearest Neighbours Are Distributed
Consider any atom in the FCC crystal — most clearly visualised as a face-centre atom. Its 12 nearest neighbours are distributed in three groups of 4:
- 4 neighbours in the same {111} close-packed plane: The atom lies in a hexagonal close-packed layer. Within this layer, it is surrounded by 6 atoms, but 4 of these are at the nearest-neighbour distance.
- 4 neighbours in the {111} plane above: The layer above fits into the triangular hollows of the current layer, placing 4 atoms at the nearest-neighbour distance directly above.
- 4 neighbours in the {111} plane below: Symmetrically, 4 atoms at the same distance in the layer directly below.
Total: 4 + 4 + 4 = 12 nearest neighbours. CN = 12.
The nearest-neighbour distance in FCC (the actual touching distance between atom centres) is:
Nearest-neighbour distance = a/√2 = a√2/2 = 2R (atoms touching — confirms hard sphere model)
It is a mathematical theorem — proved by the Kepler conjecture, formally resolved in 1998 — that no arrangement of identical spheres can pack more efficiently than 74.05%, and that in any arrangement achieving this maximum, every sphere touches exactly 12 others. FCC and HCP both achieve this theoretical maximum. No cubic or hexagonal crystal structure has CN higher than 12.
6. Atomic Packing Factor — Complete Derivation
The APF measures what fraction of the unit cell volume is actually occupied by atoms. The derivation for FCC is one of the most important calculations in introductory materials science, as covered in detail by Callister and Rethwisch — Materials Science and Engineering.
Step 1: Number of atoms
N = 4 atoms per FCC unit cell
Step 2: Volume of atoms in unit cell
V_atoms = N × (4/3)πR³ = 4 × (4/3)πR³ = (16/3)πR³
Step 3: Express unit cell volume in terms of R
From 4R = a√2: a = 4R/√2 = 2√2 R
V_cell = a³ = (2√2 R)³ = 8 × 2√2 × R³ = 16√2 R³
Step 4: Calculate APF
APF = V_atoms / V_cell
= [(16/3)πR³] / [16√2 R³]
= (16π) / (3 × 16√2)
= π / (3√2)
APF (FCC) = π/(3√2) = 3.14159 / (3 × 1.4142) = 3.14159 / 4.2426 = 0.7405
The FCC structure achieves the theoretical maximum packing efficiency of 74.05% — 6% more than BCC (68.02%) and 22% more than Simple Cubic (52.36%). The remaining 25.95% of the FCC unit cell volume is empty interstitial space, distributed in octahedral and tetrahedral voids.
7. ABCABC Stacking — The Geometry Behind FCC Close-Packing
One of the most elegant aspects of the FCC structure — and one that is frequently underexplained in introductory courses — is how its unit cell geometry connects to the close-packed layer stacking sequence.
7.1 Close-Packed Planes in FCC
The {111} planes of the FCC crystal are the most densely packed planes — each atom in these planes is surrounded by 6 nearest neighbours within the plane, arranged in a perfect hexagonal pattern. When you stack multiple {111} planes, atoms in each new layer nestle into the triangular hollows of the layer below.
Close-packed layers can stack in two distinct ways. If Layer A defines the base, and Layer B fills hollows of Layer A, then the third layer can fill one of two sets of hollows:
- ABCABC... (FCC stacking): The third layer C is positioned above hollows not occupied by Layer A atoms. The 4th layer then repeats A. This 3-layer periodicity defines the FCC structure. The {111} planes are the close-packed planes.
- ABABAB... (HCP stacking): The third layer returns directly above Layer A atoms. This 2-layer periodicity defines the HCP structure.
Both achieve the same packing efficiency (74.05%) and the same coordination number (12). The difference lies in the stacking sequence — and this difference in geometry creates the different slip systems and mechanical behaviours of FCC vs HCP metals.
This stacking perspective also reveals why FCC has 4 {111} planes (not just 1): the cube has 4 body diagonals, and each defines an independent {111} plane orientation. This is what gives FCC its 12 slip systems (4 planes × 3 directions each).
8. Slip Systems — Why FCC Metals Are Exceptionally Ductile
When a metallic crystal deforms plastically under stress, atomic layers slide over one another along specific crystallographic planes and directions. Each combination of a slip plane and a slip direction is called a slip system. The number of available slip systems determines how easily a material can deform plastically — and therefore how ductile it is.
8.1 FCC Slip Planes and Directions
In FCC, plastic deformation occurs along the most densely packed planes and directions because these offer the lowest energy barrier for dislocation glide:
- Slip planes: {111} — the close-packed planes. There are 4 independent {111} planes in the cubic system: (111), (1̄11), (11̄1), and (111̄).
- Slip directions: ⟨110⟩ — the close-packed directions within each {111} plane. Each {111} plane contains 3 independent ⟨110⟩ directions.
Total FCC slip systems = 4 planes × 3 directions per plane = 12 slip systems
8.2 Why 12 Slip Systems Means Maximum Ductility
The von Mises criterion states that a polycrystalline material requires at least 5 independent slip systems to undergo arbitrary plastic deformation without fracture. With 12 slip systems, FCC metals far exceed this threshold. No matter which direction you apply stress to an FCC polycrystal, multiple slip systems are simultaneously activated, allowing the crystal to accommodate deformation in all directions.
- FCC: 12 slip systems {111}⟨110⟩ → highest ductility — Cu, Al, Au can be drawn into fine wires, rolled into foils, stamped into complex shapes
- BCC: Up to 48 slip systems geometrically, but slip planes are not close-packed → moderate ductility, ductile-to-brittle transition at low temperatures
- HCP: Only 3 primary slip systems (basal) → limited ductility, most HCP metals are brittle at room temperature
This is why copper wire can be drawn to diameters below 0.1 mm without fracturing, why gold leaf can be beaten to thicknesses of 100 nm, and why aluminium can be rolled into wrapping foil thinner than a human hair. The FCC crystal structure, through its 12 slip systems, is the geometric reason for all of this.
9. Interstitial Voids in the FCC Structure
Even with 74.05% packing efficiency, the FCC structure retains significant interstitial space. The geometry of these void spaces is important for understanding solid solution strengthening, diffusion kinetics, and the behaviour of interstitial atoms (H, C, N, Li) in FCC metals.
9.1 Octahedral Interstitial Sites
An octahedral void is surrounded by 6 FCC atoms arranged at the vertices of a regular octahedron. In the FCC unit cell, octahedral voids are located at:
- The body centre of the unit cell: position (½,½,½) — 1 site entirely inside the cell
- The midpoints of all 12 edges of the cube, each shared among 4 unit cells: 12 × (1/4) = 3 sites
Octahedral voids per FCC unit cell = 1 + 3 = 4
Octahedral void radius ratio: r/R = 0.414
(Maximum atom fitting without distortion: r = 0.414 R)
Carbon atoms (radius ~77 pm) occupy octahedral interstitial sites in FCC gamma-iron (austenite, R_Fe = 126 pm). The octahedral void radius in FCC iron is 0.414 × 126 = 52 pm. While carbon atoms are still larger than this void, the distortion is manageable — carbon can dissolve up to 2.14 wt% in austenite. This is 100× more than in BCC alpha-iron (0.022 wt%), because BCC octahedral voids (r/R = 0.154) are far smaller. This difference is the physical basis of the entire science of steel metallurgy and heat treatment.
9.2 Tetrahedral Interstitial Sites
Tetrahedral voids are surrounded by 4 FCC atoms arranged at the vertices of a tetrahedron. In the FCC unit cell, there are 8 tetrahedral voids — all located inside the cell at positions like (¼,¼,¼).
Tetrahedral voids per FCC unit cell = 8
Tetrahedral void radius ratio: r/R = 0.225
(Smaller than octahedral voids — only very small atoms fit)
| Void Type | Number per FCC Unit Cell | Location | Radius ratio r/R | Example occupant |
|---|---|---|---|---|
| Octahedral | 4 | Body centre + edge midpoints | 0.414 | C in austenite, Li in Cu |
| Tetrahedral | 8 | Interior at (¼,¼,¼) positions | 0.225 | H in Pd, small interstitials |
The ratio of interstitial sites to FCC atoms is 4 octahedral : 4 atoms = 1 octahedral site per atom, and 8 tetrahedral : 4 atoms = 2 tetrahedral sites per atom. This has profound implications for lithium-ion battery cathode materials, hydrogen storage alloys, and carburisation processes in steel manufacture.
10. Theoretical Density of FCC Metals
Using the standard density formula with n = 4 for FCC:
ρ = (n × A) / (V_c × N_A) = (4 × A) / (a³ × N_A)
Where:
n = 4 (FCC — always)
A = atomic mass (g/mol)
a = lattice parameter in cm (1 nm = 10⁻⁷ cm)
N_A = 6.022 × 10²³ mol⁻¹
Lattice parameters in crystallographic databases are given in nm or Å. Convert to cm before calculating density:
1 nm = 10⁻⁷ cm | 1 Å = 10⁻⁸ cm
Example: a(Cu) = 0.3615 nm = 3.615 × 10⁻⁸ cm → a³ = (3.615)³ × 10⁻²⁴ = 47.27 × 10⁻²⁴ cm³
11. Common FCC Metals and Engineering Applications
| Metal | Symbol | a (nm) | R (pm) | ρ (g/cm³) | Key Engineering Applications |
|---|---|---|---|---|---|
| Aluminium | Al | 0.4050 | 143 | 2.70 | Aerospace, automotive, packaging, transmission lines |
| Copper | Cu | 0.3615 | 128 | 8.94 | Electrical wiring, PCBs, heat exchangers, plumbing |
| Gold | Au | 0.4078 | 144 | 19.30 | Electronic connectors, bonding wires, corrosion-resistant contacts |
| Silver | Ag | 0.4086 | 144 | 10.49 | Photography, mirrors, antimicrobial surfaces, solar cells |
| Nickel | Ni | 0.3524 | 125 | 8.91 | Superalloys (jet engines), batteries (NiMH), plating |
| Lead | Pb | 0.4950 | 175 | 11.34 | Radiation shielding, batteries, solders |
| Platinum | Pt | 0.3924 | 139 | 21.45 | Catalytic converters, fuel cells, jewellery |
| Palladium | Pd | 0.3890 | 137 | 12.02 | Hydrogen storage, catalysis, electronics |
Iron undergoes allotropic transformation: below 912°C it is BCC (alpha-iron, ferrite), between 912°C and 1394°C it is FCC (gamma-iron, austenite), and above 1394°C it returns to BCC (delta-iron). The FCC gamma-iron phase is crucial for steel heat treatment — it can dissolve up to 2.14 wt% carbon (vs 0.022 wt% in BCC alpha-iron), enabling the formation of martensite during quenching. Without the BCC↔FCC transformation in iron, the entire technology of steel heat treatment — and the structural steel industry — would not exist.
12. FCC vs BCC vs HCP — Master Comparison
| Property | FCC | BCC | HCP |
|---|---|---|---|
| Atoms per unit cell | 4 | 2 | 6 |
| Coordination number | 12 | 8 | 12 |
| Touching direction | Face diagonal [110] | Body diagonal [111] | Basal plane edge [1120] |
| a vs R | a = 2√2 R | a = 4R/√3 | a = 2R |
| APF (exact) | π/(3√2) | π√3/8 | π/(3√2) |
| APF (%) | 74.05% | 68.02% | 74.05% |
| Void fraction | 25.95% | 31.98% | 25.95% |
| Octahedral void r/R | 0.414 | 0.154 | 0.414 |
| Stacking sequence | ABCABC {111} | Not close-packed | ABABAB (0001) |
| Slip systems | 12 {111}⟨110⟩ | Up to 48 | 3 basal (primary) |
| Ductility | Highest | Moderate | Low–Moderate |
| DBTT | No | Yes | Partial |
| C solubility | 2.14 wt% (austenite) | 0.022 wt% (ferrite) | Varies |
| Example metals | Cu, Al, Au, Ag, Ni | Fe, Cr, W, Mo, V | Mg, Ti, Zn, Co |
13. Advanced Frontier: FCC in Nanomaterials and Quantum Devices (2025)
The FCC crystal structure continues to underpin cutting-edge research in 2025–2026, demonstrating that understanding this classical structure opens doors to entirely new physics.
13.1 FCC Gold and Silver Nanoparticles
Metallic nanoparticles of gold, silver, and platinum maintain their FCC crystal structure down to diameters of ~2 nm (containing only a few hundred atoms), but their properties diverge dramatically from bulk FCC metals. At the nanoscale, the surface-to-volume ratio becomes enormous, and a large fraction of atoms occupy surface sites with reduced coordination number. This creates quantum confinement effects, surface plasmon resonance, and exceptional catalytic activity. Gold nanoparticles exhibiting FCC structure are used as contrast agents in biological imaging, photothermal cancer therapy agents, and surface-enhanced Raman spectroscopy (SERS) substrates.
13.2 High-Entropy Alloys — Multi-Principal FCC Structures
A recent revolution in alloy design is the development of high-entropy alloys (HEAs) — alloys containing five or more principal elements in near-equimolar proportions. Remarkably, many of these complex, chemically disordered alloys adopt a single FCC crystal structure, despite their chemical complexity. The celebrated CrMnFeCoNi Cantor alloy (equimolar, all elements) adopts a single-phase FCC structure and exhibits exceptional fracture toughness — including at cryogenic temperatures where BCC steels become brittle. Research published in Science (2014) by Gludovatz et al. demonstrated that this FCC HEA maintains toughness exceeding 200 MPa·m^½ at liquid nitrogen temperatures — significantly better than most conventional alloys.
13.3 FCC Topological Metals
Several newly discovered topological quantum materials adopt FCC crystal structures. The Weyl semimetals and topological nodal-line semimetals in the FCC family have linear band crossings protected by the crystal symmetry of the FCC lattice — particularly its time-reversal and inversion symmetries. The ability to predict and design such materials relies entirely on understanding the FCC reciprocal lattice (its first Brillouin zone is a truncated octahedron) and how electronic states transform under the FCC symmetry operations.
14. Solved Numerical Problems
For FCC, atoms touch along the face diagonal: 4R = a√2
R = a√2 / 4 = (0.4078 × 1.4142) / 4 = 0.5767 / 4 = 0.1442 nm = 144.2 pm ✔
Tabulated atomic radius of Au = 144 pm — excellent agreement.
n = 4 (FCC)
a = 0.4050 nm = 4.050 × 10⁻⁸ cm
V_c = a³ = (4.050 × 10⁻⁸)³ = 66.43 × 10⁻²⁴ cm³
ρ = (4 × 26.98) / (66.43 × 10⁻²⁴ × 6.022 × 10²³)
= 107.92 / 40.00
= 2.698 g/cm³ ✔
Experimentally measured density of Al = 2.699 g/cm³ — essentially exact agreement.
For FCC octahedral void: r_void = 0.414 × R_Fe
r_void = 0.414 × 126 pm = 52.2 pm
Carbon atomic radius = 77 pm > 52.2 pm
Carbon is larger than the undistorted octahedral void by a factor of:
77 / 52.2 = 1.48 (48% larger than the void)
But the FCC lattice is relatively elastic and accommodates this distortion,
allowing up to 2.14 wt% C dissolution in austenite. ✔
This calculation explains why carbon dissolves in austenite with significant lattice strain, contributing to the hardening of steel during rapid cooling (quenching).
Step 1: N = 4 atoms per FCC unit cell
Step 2: V_atoms = 4 × (4/3)πR³ = (16/3)πR³
Step 3: 4R = a√2 → a = 2√2 R → a³ = (2√2)³ R³ = 16√2 R³
Step 4: APF = (16/3)πR³ / (16√2 R³)
= (16π/3) / (16√2)
= π / (3√2)
= 3.14159 / 4.24264
= 0.7405 = 74.05% ✔
15. Practice Multiple Choice Questions
- (a) Cube edge ⟨100⟩
- (b) Body diagonal ⟨111⟩
- (c) Face diagonal ⟨110⟩ ✔ — giving 4R = a√2
- (d) They do not touch in any direction
- (a) π/6 (= 52.36% — Simple Cubic)
- (b) π√3/8 (= 68.02% — BCC)
- (c) π/(3√2) = 0.7405 ✔ — the theoretical maximum for equal spheres
- (d) π/4 (= 78.54%)
- (a) FCC iron is at higher temperature, so carbon is more mobile
- (b) FCC octahedral voids have r/R = 0.414, much larger than BCC octahedral voids (r/R = 0.154) ✔
- (c) FCC iron has a larger lattice parameter than BCC iron
- (d) Carbon forms covalent bonds with FCC iron atoms
- (a) 2
- (b) 4 ✔ — 1 body-centre + 12 edge-midpoints × (1/4) = 1 + 3 = 4
- (c) 6
- (d) 8
- (a) Body-diagonal touching direction
- (b) Only 3 independent slip systems
- (c) 4 independent {111} slip planes, giving 12 total slip systems and exceptional ductility ✔
- (d) A ductile-to-brittle transition temperature similar to BCC
- (a) More brittle, because FCC has higher packing
- (b) About the same toughness as steel
- (c) Significantly tougher — FCC metals lack a DBTT and maintain ductility at liquid nitrogen temperatures ✔
- (d) Only ductile if the composition is adjusted to pure iron
16. Key Takeaways
- FCC DEFINITION: Cubic unit cell with atoms at all 8 corners and the centre of each of the 6 faces. Belongs to the cubic crystal system (a = b = c, α = β = γ = 90°).
- ATOMS/CELL: N = 8×(1/8) + 6×(1/2) = 4 atoms per unit cell — the highest of any simple cubic-type structure.
- TOUCHING DIRECTION: Face diagonal ⟨110⟩. Along this diagonal: corner (R) + face-centre (2R) + corner (R) = 4R = a√2.
- a vs R: a = 2√2 R = 2.828 R | R = a√2/4 = 0.3536 a.
- COORDINATION NUMBER: CN = 12 — the theoretical maximum for equal spheres. 4 neighbours in the same {111} plane, 4 above, 4 below.
- APF: 74.05% = π/(3√2) — the theoretical maximum packing efficiency (Kepler conjecture). Void fraction = 25.95%.
- STACKING: ABCABC... sequence of {111} close-packed planes. This 3-layer periodicity defines the FCC structure and distinguishes it from HCP (ABABAB).
- SLIP SYSTEMS: 12 slip systems — {111}⟨110⟩ (4 planes × 3 directions). This explains the exceptional ductility of FCC metals and absence of DBTT.
- INTERSTITIAL VOIDS: 4 octahedral voids (r/R = 0.414) and 8 tetrahedral voids (r/R = 0.225) per unit cell. Large octahedral voids accommodate carbon in austenite (2.14 wt%).
- DENSITY: ρ = (4 × A) / (a³ × N_A). With n = 4 always for FCC.
- COMMON METALS: Cu, Al, Au, Ag, Ni, Pb, Pt, Pd — plus gamma-iron between 912–1394°C. All highly ductile, electrically conductive, formable.
- 2025 FRONTIER: FCC high-entropy alloys (Cantor alloy) offer exceptional cryogenic toughness. FCC gold nanoparticles enable biomedical imaging and cancer therapy.
Series Navigation
References
All references are in IEEE citation style. All sources are peer-reviewed journals, internationally recognised textbooks, or authoritative academic databases.
- W. D. Callister Jr. and D. G. Rethwisch, Materials Science and Engineering: An Introduction, 10th ed. Hoboken, NJ, USA: John Wiley & Sons, 2018, ch. 3, pp. 40–78. [Wiley] — Primary textbook reference for FCC unit cell geometry, atom counting, APF derivation, slip systems, and interstitial void analysis.
- C. Kittel, Introduction to Solid State Physics, 8th ed. Hoboken, NJ, USA: John Wiley & Sons, 2005, ch. 1, pp. 1–26. [Wiley] — Reference for FCC Bravais lattice, primitive vectors, ABCABC stacking, and first Brillouin zone of the FCC lattice.
- N. W. Ashcroft and N. D. Mermin, Solid State Physics. Philadelphia, PA, USA: Holt, Rinehart and Winston, 1976, ch. 4–5. — Graduate reference for FCC crystal structure, close-packed planes, and electronic properties arising from FCC symmetry.
- A. Kelly and K. M. Knowles, Crystallography and Crystal Defects, 2nd ed. Chichester, UK: John Wiley & Sons, 2012. [Wiley] — Reference for FCC slip systems, interstitial void geometry, and dislocation behaviour in FCC metals.
- T. H. Hales et al., "A formal proof of the Kepler conjecture," Forum Math. Pi, vol. 5, Art. no. e2, 2017, doi: 10.1017/fmp.2017.1. [Cambridge University Press — Open Access DOI] — Formal mathematical proof that 74.05% is the maximum packing efficiency for equal spheres — confirming FCC (and HCP) achieve the theoretical maximum.
- B. Gludovatz et al., "A fracture-resistant high-entropy alloy for cryogenic applications," Science, vol. 345, no. 6201, pp. 1153–1158, Sep. 2014, doi: 10.1126/science.1254581. [Science — DOI: 10.1126/science.1254581] — Landmark paper demonstrating exceptional cryogenic fracture toughness of the single-phase FCC Cantor alloy CrMnFeCoNi.
- A. Jain et al., "Commentary: The Materials Project: A materials genome approach to accelerating materials innovation," APL Mater., vol. 1, no. 1, Art. no. 011002, 2013, doi: 10.1063/1.4812323. [Materials Project — Open Crystal Database] — Source for all FCC metal lattice parameters, densities, and crystal structure data in Table 1 of this lecture.
- University of Cambridge, DoITPoMS, "Iron-Carbon Phase Diagram," Teaching and Learning Packages. Cambridge, UK: University of Cambridge, 2023. [doitpoms.ac.uk] — Reference for carbon solubility in FCC austenite (2.14 wt%) vs BCC ferrite (0.022 wt%) and the role of FCC structure in steel metallurgy.
- R. Verma and S. K. Rout, "Frequency-dependent ferro–antiferro phase transition and internal bias field influenced piezoelectric response of donor and acceptor doped bismuth sodium titanate ceramics," J. Appl. Phys., vol. 126, no. 9, Art. no. 094103, Sep. 2019, doi: 10.1063/1.5111505. [AIP — DOI: 10.1063/1.5111505] — Author's peer-reviewed research on crystal structure and phase analysis in perovskite materials, relevant to the discussion of iron's FCC↔BCC allotropic transformation and its functional implications.
- R. Verma and S. K. Rout, "The Mystery of Dimensional Effects in Ferroelectricity," in Recent Advances in Multifunctional Perovskite Materials. London, UK: IntechOpen, 2022, doi: 10.5772/intechopen.104435. [IntechOpen — Open Access] — Author's chapter on structural transitions in perovskite materials, relevant to the FCC structure of high-temperature phases in functional oxides.
This tutorial is part of the Crystal Structure Hub Series at AdvanceMaterialsLab.com
Next in series: HCP Crystal Structure — Complete Analysis →
© 2026 AdvanceMaterialsLab.com — Dr. Rolly Verma | All rights reserved
1️⃣ FCC Unit Cell & Atomic Radius Numericals
Problem 1 (GATE Level)
Aluminum crystallizes in an FCC structure. The atomic radius is 143 pm. Find the lattice parameter.
Solution
For FCC:
a = 2√2 R
Substitute
a = 2√2 × 143 a = 2 × 1.414 × 143 a = 404.4 pm
Final Answer
a ≈ 4.04 Å
Problem 2 (NET Level)
An FCC crystal has lattice parameter a = 4.0 Å. Find the atomic radius.
Solution
a = 2√2 R R = a / (2√2)
R = 4 / (2 × 1.414)
R = 1.414 Å
Final Answer
R = 1.41 Å
2️⃣ Number of Atoms in FCC Numericals
Problem 3
Calculate the number of atoms present in one FCC unit cell.
Solution
Corner atoms
8 × 1/8 = 1
Face atoms
6 × 1/2 = 3
Total atoms
Z = 1 + 3 = 4
Final Answer
Z = 4
Problem 4 (Conceptual Numerical)
How many atoms are present in 10 FCC unit cells?
Solution
Z = 4
10 × 4 = 40
Final Answer
40 atoms
3️⃣ Density Numericals (Very Common in GATE)
Problem 5
Copper crystallizes in an FCC structure.
Atomic weight M = 63.5 g/mol
Lattice parameter a = 3.61 Å
Find the density.
Formula
ρ = ZM / (Nₐ a³)
Where Z = 4
Convert lattice parameter
a = 3.61 × 10⁻⁸ cm
Substitute
ρ = (4 × 63.5) / [6.022 × 10²³ × (3.61 × 10⁻⁸)³]
Final Answer
ρ ≈ 8.96 g/cm³
Problem 6
An FCC metal has lattice parameter a = 4.05 Å
Atomic weight M = 108 g/mol
Find the density.
Solution
ρ = ZM / (Nₐ a³)
Z = 4
a = 4.05 × 10⁻⁸ cm
ρ = (4 × 108) / [6.022 × 10²³ × (4.05 × 10⁻⁸)³]
Final Answer
ρ ≈ 10.5 g/cm³
4️⃣ Coordination Number Numericals
Problem 7
Determine the coordination number of FCC.
Solution
Each atom is surrounded by:
- 4 atoms in the same plane
- 4 atoms above
- 4 atoms below
4 + 4 + 4 = 12
Final Answer
12
5️⃣ Atomic Packing Factor Problems
Problem 8
Calculate the packing efficiency of FCC structure.
Solution
Volume of atoms
4 × (4/3)πR³
Unit cell volume
a³
For FCC
a = 2√2 R
Substitute
APF = π / (3√2)
Final Answer
APF = 0.74
Packing efficiency
74%
6️⃣ Void Numericals (Common in NET)
Problem 9
Find the number of tetrahedral voids in FCC.
Solution
Tetrahedral voids = 2Z
Z = 4
2 × 4 = 8
Final Answer
8
Problem 10
Find the number of octahedral voids in FCC.
Solution
Octahedral voids = Z
Z = 4
Final Answer
4
7️⃣ Higher-Level Numerical (NET Type)
Problem 11
Calculate the edge length of an FCC unit cell if atomic radius R = 1.28 Å.
Solution
a = 2√2 R
a = 2 × 1.414 × 1.28
a = 3.62 Å
8️⃣ Reverse Density Problem (GATE Style)
Problem 12
An FCC crystal has density 8 g/cm³.
Atomic weight 64 g/mol.
Find the lattice parameter.
Solution
ρ = ZM / (Nₐ a³)
Solve for a
a = (ZM / ρNₐ)^(1/3)
9️⃣ Conceptual Numerical
Problem 13
What is the packing efficiency difference between FCC and BCC?
Solution
FCC = 74%
BCC = 68%
Difference = 6%
🔟 Crystal Plane Numerical
Problem 14
Determine the number of atoms touching one atom in FCC.
Answer
12
Frequently Asked Questions (FAQs)
-
Why do many metals prefer the FCC crystal structure?
Many metals adopt the Face-Centered Cubic (FCC) structure because it provides high packing efficiency and energetic stability. The FCC lattice allows atoms to arrange themselves in a closely packed configuration, minimizing the total energy of the system. In addition, the FCC structure contains multiple slip systems that enable metals to deform easily without fracturing.
-
Why does the FCC structure have the highest ductility among cubic crystal systems?
The FCC structure possesses 12 independent slip systems, which allow atoms to move along several crystallographic planes and directions when stress is applied. Because deformation can occur along multiple paths, FCC metals such as copper and aluminium can undergo significant plastic deformation before failure. This is why FCC metals are generally more ductile than BCC or HCP metals.
-
How does the FCC crystal structure influence diffusion in metals?
In FCC crystals, diffusion often occurs through vacancies and interstitial sites within the lattice. The presence of relatively large octahedral interstitial sites allows smaller atoms to migrate through the structure more easily. Diffusion behavior is important in processes such as alloy formation, heat treatment, and phase transformations.
-
Why is the atomic packing factor of FCC equal to 0.74?
The Atomic Packing Factor (APF) of the FCC structure is 0.74 because atoms are arranged in a close-packed configuration where spheres occupy the maximum possible space without overlapping. In this arrangement, atoms touch along the face diagonal of the cube, resulting in highly efficient packing where about 74% of the unit cell volume is filled with atoms.
-
How does the FCC structure influence electrical conductivity in metals?
In FCC metals such as copper and silver, the ordered atomic arrangement allows electrons to move relatively freely through the lattice. Metallic bonding and the regular structure facilitate efficient electron transport, which is why FCC metals are widely used in electrical conductors and electronic devices.
-
Why are FCC nanoparticles important in nanotechnology?
Many metallic nanoparticles including gold, silver, platinum, and palladium adopt the FCC structure. At the nanoscale, crystallographic surfaces such as the (111) and (100) planes strongly influence surface energy, catalytic activity, and chemical reactions, making FCC nanoparticles important in catalysis, sensing technologies, and biomedical applications.
-
How do crystal defects affect FCC materials?
Although the FCC structure is highly ordered, real materials contain defects such as vacancies, dislocations, and grain boundaries. These defects influence mechanical properties by controlling dislocation motion, plastic deformation, and the strength and stability of the material.
-
Why are FCC metals widely used in engineering applications?
FCC metals combine several desirable properties such as high ductility, good electrical conductivity, corrosion resistance, and excellent formability. Because of these advantages, they are widely used in aerospace components, electrical wiring, electronic devices, nanomaterials, and advanced structural materials.
Dr. Rolly Verma
If you notice any inaccuracies or have constructive suggestions to improve the content, I warmly welcome your feedback. It helps maintain the quality and clarity of this educational resource. You can reach me at: advancematerialslab27@gmail.com
Explore More from Advance Materials Lab
Continue expanding your understanding of material behavior through our specialized resources on ferroelectrics and phase transitions. Each guide is designed for research scholars and advanced learners seeking clarity, precision, and depth in the field of materials science and nanotechnology.
🔗 Recommended Reads and Resources:
Ferroelectrics Tutorials and Research Guides — Comprehensive tutorials covering polarization, hysteresis, and ferroelectric device characterization.
Workshops on Ferroelectrics (2025–2027) — Upcoming training sessions and research-oriented workshops for hands-on learning.
Glossary — Ferroelectrics and Phase Transitions — Concise explanations of key terminologies to support your study and research work.
