LECTURE 04
Hexagonal Close-Packed (HCP) Crystal Structure
A Complete Structural Analysis for Materials Science Students
By Dr. Rolly Verma | AdvanceMaterialsLab.com | Published: March 2026 | B.Sc. / M.Sc. Materials Science & Engineering
Topic: Hexagonal Close-Packed (HCP) Crystal Structure | Level: Undergraduate / Postgraduate
Estimated reading time: 30 minutes | Includes: Theory, Derivations, Tables, MCQs, Solved Problems
SEO Keywords: HCP crystal structure, hexagonal close-packed, atomic packing factor HCP, coordination number HCP, c/a ratio, HCP vs FCC, slip systems HCP, HCP metals, crystal lattice, materials science tutorial
1. Introduction: Setting the Stage
Welcome to this tutorial. If you have already studied the Body-Centred Cubic (BCC) and Face-Centred Cubic (FCC) structures, you are now ready to encounter the third great member of the crystal structure family — the Hexagonal Close-Packed (HCP) structure. If this is your first encounter with crystal structures, do not worry at all. We will build every idea from the ground up, one brick at a time.
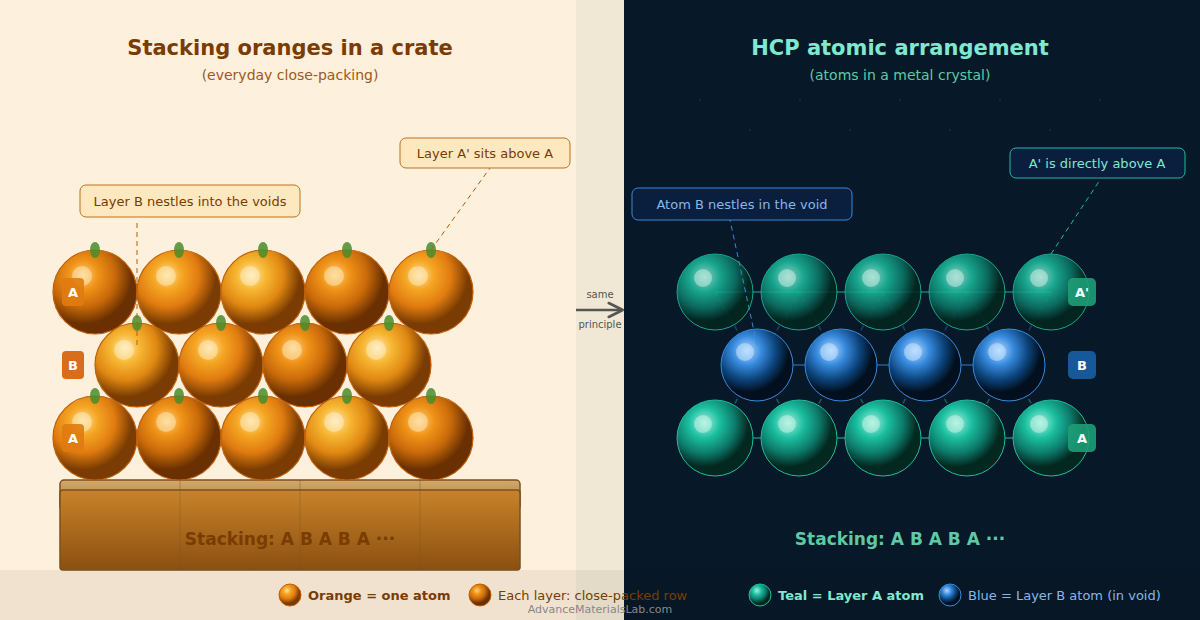
Let us start with a simple question. Have you ever stacked oranges in a fruit basket? If you have, you may have noticed that the most natural way to stack them is to first lay them in a flat layer, and then nestle the second layer in the dips between the oranges of the first layer. This gives the most compact arrangement possible. Nature does exactly the same thing with atoms in certain metals, and the result is the Hexagonal Close-Packed (HCP) crystal structure.
HCP is one of the two ways to achieve the densest possible packing of identical spherical atoms — the other being the Face-Centred Cubic (FCC) structure. Both achieve the same theoretical maximum packing efficiency of 74%. The difference between them is subtle but profoundly important: it lies in how the close-packed layers are stacked on top of each other. This single difference in stacking pattern leads to different slip systems, different plastic deformation behaviours, and ultimately different engineering properties.
HCP is not merely an academic concept. Magnesium — the lightest structural metal — is HCP. Titanium — the backbone of the aerospace industry — is HCP at room temperature. Zinc — used to protect steel from corrosion — is HCP. Understanding HCP is therefore essential for anyone working in lightweight structures, biomedical implants, aerospace alloys, or advanced coatings.
2. Crystalline Solids and the Concept of a Lattice
2.1 What Makes a Solid 'Crystalline'?
In materials science, we broadly classify solids into two categories: crystalline and amorphous. In a crystalline solid, the atoms, ions, or molecules are arranged in a perfectly ordered, repeating three-dimensional pattern that extends over the entire material. This long-range order is what distinguishes a crystalline solid from a glass or an amorphous material, where there is only short-range or no order at all.
Think of a tiled floor. If the tiles are arranged in a perfectly regular geometric pattern — say, identical square tiles in neat rows and columns — it is analogous to a crystalline solid. If the tiles are placed randomly with no repeating pattern, it is analogous to an amorphous solid. Most metals and ceramics that we use in engineering are crystalline in nature.
2.2 The Space Lattice and the Unit Cell
To describe a crystal mathematically, we use the concept of a space lattice — an infinite array of points in space where each point has an identical environment. The space lattice is an abstract geometric framework; atoms or groups of atoms are then associated with each lattice point.
From this infinite lattice, we identify the smallest repeating unit, called the unit cell. The unit cell is the fundamental building block of the crystal. By translating (repeating) the unit cell in three dimensions, we can reproduce the entire crystal structure. Think of the unit cell as a single LEGO brick — one brick looks simple, but stacking millions of identical bricks in all directions creates the entire structure.
A unit cell is completely described by six parameters: three edge lengths (a, b, c) and three angles (α, β, γ) between those edges. These six parameters are known as the lattice parameters. For the hexagonal system, the relevant parameters are the in-plane edge length a and the height c (the vertical edge of the hexagonal prism).
- Crystal lattice: An infinite, ordered, repeating arrangement of atoms in three-dimensional space.
- Unit cell: The smallest repeating structural unit of a crystal, from which the entire crystal can be reproduced by translation.
- Lattice parameters: The six quantities (a, b, c, α, β, γ) that completely describe the geometry of a unit cell.
- Basis: The atom or group of atoms associated with each lattice point.
3. Building the HCP Structure: A Step-by-Step Construction
This is the heart of our tutorial. We are going to construct the HCP structure layer by layer, as if we were actually placing atoms in space. By the time we finish, you will have a clear mental image of exactly how the atoms are arranged and why this arrangement is called 'close-packed'.
3.1 Step 1 — The Close-Packed Layer (Layer A)
Begin by placing identical spherical atoms (think of them as billiard balls) on a flat surface. Arrange them so that each atom touches as many neighbours as possible. The most efficient way to do this on a flat surface is to arrange them in rows, and then offset alternate rows by half a ball diameter so that balls in the second row nestle into the gaps of the first row.
The result is a hexagonally arranged layer — when you look at any single atom, it is surrounded by exactly 6 others at the same level. Each atom touches all 6 neighbours in the plane. This is the close-packed layer, and it has the highest possible packing density for a single flat layer. We call this Layer A.
Notice the geometry: the 6 surrounding atoms form a regular hexagon around the central atom. The centre-to-centre distance between any two touching atoms equals 2R, where R is the atomic radius. Equivalently, this distance equals the lattice parameter a. So in HCP, a = 2R always.
Imagine laying oranges on a flat table in the most space-efficient way. Each orange sits in the gap between two adjacent oranges in the row below. From above, you see a hexagonal pattern — each orange surrounded by 6 others. This is your Layer A. Everything that follows in HCP is simply about how you stack the next layer on top.
3.2 Step 2 — Identifying the Voids Above Layer A
Now look at Layer A from above. Between the atoms, you will notice two types of triangular gaps:
- Type 1 (B-sites): Triangular gaps where the three surrounding atoms have their vertices pointing upward — these look like upward-pointing triangles from above.
- Type 2 (C-sites): Triangular gaps where the three surrounding atoms have their vertices pointing downward — downward-pointing triangles from above.
In a close-packed layer, Type 1 (B) and Type 2 (C) voids alternate across the entire layer. The key question that distinguishes HCP from FCC is: which set of sites do we use for the second layer? For the HCP structure, we will use the B-sites for the middle layer.
3.3 Step 3 — Placing Layer B (The Middle Layer)
Now take a second set of atoms and place them above the B-sites — the upward-pointing triangular voids of Layer A. Each atom in this new layer sits directly above the centre of one of these triangular voids, nestled comfortably between three atoms of Layer A below it.
In each unit cell, there are exactly 3 atoms in the middle B layer. Each B-layer atom is simultaneously sitting in the hollow of 3 atoms from Layer A below, and it will be cradled by 3 atoms from Layer A' above — totalling 12 nearest neighbours, which is the coordination number of HCP.
3.4 Step 4 — Placing Layer A' (The Critical Step!)
Now we must place the third layer. This is where HCP and FCC diverge — the single most important structural difference between the two close-packed structures.
- Third layer placed directly above the first layer (A-sites)
- Looking straight down: Layer 3 eclipses Layer 1
- Stacking sequence: A B A B A B ···
- 2-layer periodicity
- Metals: Mg, Ti, Zn, Co, Cd
- Third layer placed above unused voids (C-sites)
- Looking straight down: all three layers are offset
- Stacking sequence: A B C A B C ···
- 3-layer periodicity
- Metals: Al, Cu, Au, Ni, Pb
In HCP, the stacking sequence repeats as A-B-A-B-A-B indefinitely. This ABABAB stacking pattern is the defining signature of the HCP structure. Despite the structural difference with FCC, both achieve the same 74% packing efficiency — the maximum theoretically possible for identical spheres.
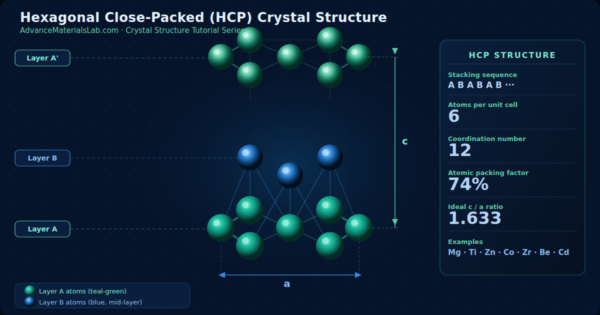
3.5 The Complete HCP Unit Cell
The HCP unit cell is a right hexagonal prism. Let us describe its geometry precisely:
- Base: A regular hexagon with edge length a (the atomic diameter in the close-packed plane).
- Height: The vertical distance c, which is the repeat distance along the stacking direction (the c-axis or [0001] direction).
- Contents: Atoms from the bottom hexagonal face, the top hexagonal face, and 3 interior atoms from Layer B.
- Lattice parameters: Defined by two quantities only: a and c. The geometry of the hexagonal close-packed structure is defined by two lattice parameters, a and c. The complete HCP unit cell can be visualized as a right hexagonal prism as shown in the diagram below.
- Shape: Regular hexagonal prism
- Edge length of hexagonal base: a (= 2R, where R = atomic radius)
- Height of prism: c
- Ideal c/a ratio: 1.633 (derived from geometry of close-packing)
- Lattice parameters: a and c (2 parameters, unlike cubic systems which need only 1)
- Stacking sequence: A B A B A B ··· (2-layer periodicity)
4. Detailed Structural Analysis of HCP
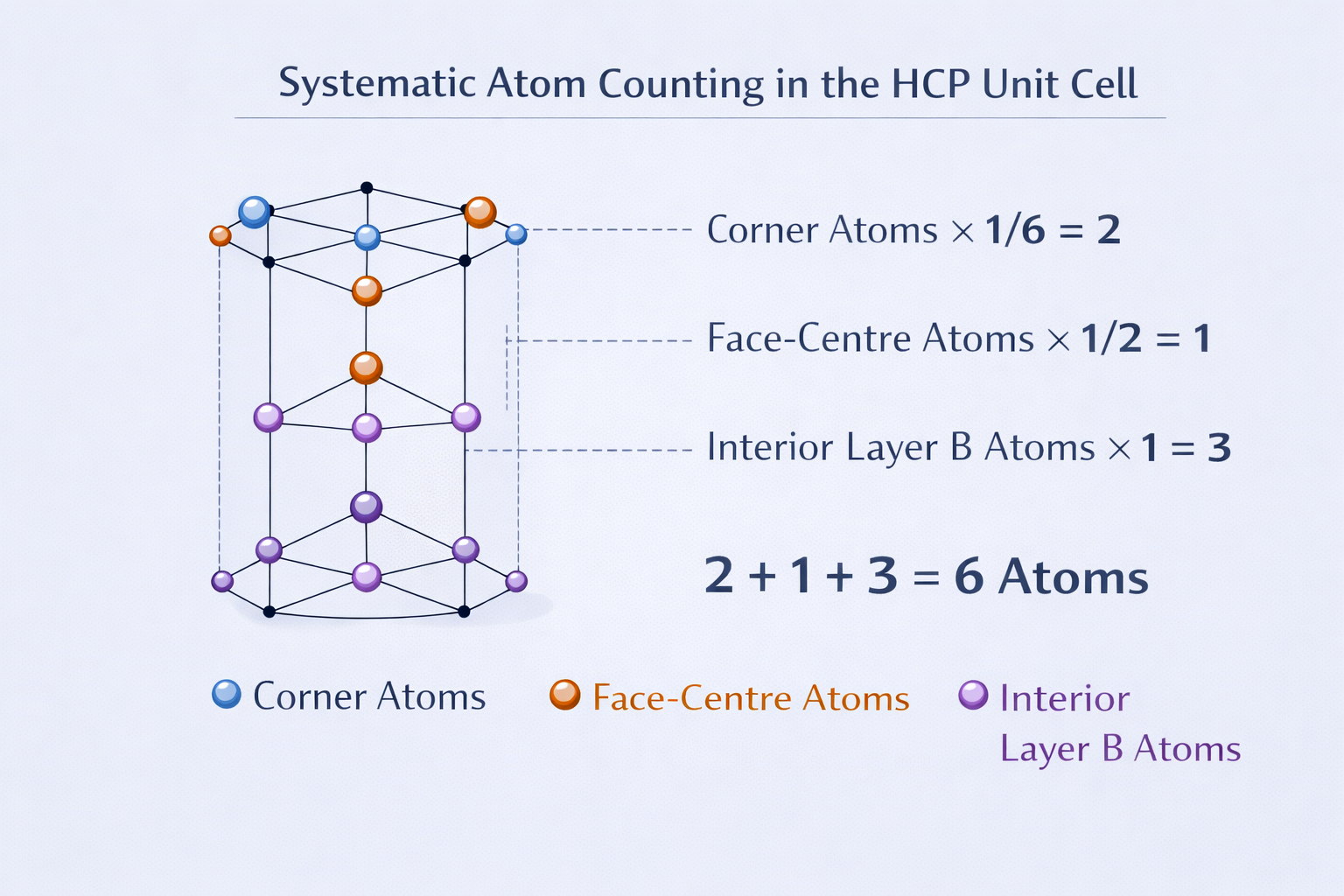
4.1 Number of Atoms per Unit Cell
This is a fundamental calculation. We need to count how many atoms effectively belong to one HCP unit cell, accounting for the fact that atoms on faces and edges are shared between adjacent unit cells.
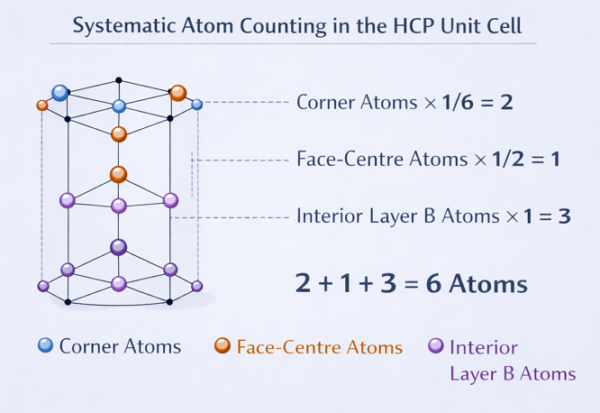
(A) Atoms at the 12 corners of the hexagonal prism
Each corner of the hexagonal prism is shared between 6 different unit cells (since 6 prisms meet at every corner vertex in a hexagonal lattice). Therefore, each corner atom contributes 1/6 of itself to our unit cell.
(B) Atoms at the 2 hexagonal face centres (top and bottom)
The centre of each hexagonal face is shared between 2 unit cells (the one above and the one below). Therefore, each face-centre atom contributes 1/2 to our unit cell.
(C) The 3 interior atoms (Layer B — middle layer)
These atoms sit entirely inside the unit cell. They are not shared with any neighbour. Each contributes fully.
Total atoms per unit cell:
This is larger than BCC (2 atoms) and FCC (4 atoms). The HCP unit cell is larger, but the packing efficiency is the same as FCC at 74%.
4.2 The Coordination Number of HCP
The coordination number (CN) is defined as the number of nearest neighbours that any given atom has — the number of atoms in direct contact with the reference atom.
Let us count the nearest neighbours of any one atom in the HCP lattice. Choose the centre atom of the bottom hexagonal face as our reference atom:
- In the same close-packed layer (Layer A): 6 nearest neighbours, all at distance a = 2R, all touching the reference atom in the same plane.
- In Layer B (middle layer, directly above): 3 nearest neighbours, each at the same distance a = 2R.
- In Layer A below (one cell down): 3 nearest neighbours, similarly at distance a = 2R.
4.3 Derivation of the Ideal c/a Ratio
This is one of the most elegant derivations in introductory materials science. Consider a central atom in Layer B and the three atoms below it in Layer A. These four atoms form a regular tetrahedron with all six edges equal to a.
Step 1: Finding the height of the tetrahedron
The distance from any vertex of an equilateral triangle of side a to its centroid is:
Using the Pythagorean theorem for the tetrahedron height h:
Step 2: Finding c from h
Since Layer B sits exactly midway between the two A layers, the full prism height c is twice h:
Step 3: The c/a ratio
This value holds only for a perfect close-packed arrangement of identical hard spheres. Real metals deviate because atoms are not perfect hard spheres and because electron bonding effects modify the interatomic spacings.
4.4 Atomic Radius vs. Lattice Parameter
In HCP, atoms in the same layer are touching. Therefore, the centre-to-centre distance equals twice the atomic radius:
4.5 Volume of the HCP Unit Cell
The unit cell is a regular hexagonal prism with base edge a and height c. The area of a regular hexagon with side a is (3√3/2)a². Therefore:
4.6 Atomic Packing Factor (APF) of HCP
The Atomic Packing Factor (APF) tells us what fraction of the unit cell volume is actually occupied by atoms — a dimensionless measure of packing efficiency.
Step 1: Volume occupied by atoms
Step 2: Volume of unit cell
Step 3: Calculate APF
This is the maximum possible packing efficiency for identical spheres in any crystal structure. Both HCP and FCC achieve this limit. By comparison, BCC achieves only 68% and simple cubic achieves only 52%. The remaining 26% of the HCP unit cell is empty space (voids or interstices).
4.7 Types of Voids in HCP: Tetrahedral and Octahedral
The 26% empty space in HCP is concentrated in well-defined geometric voids between the atoms. There are two types:
(A) Tetrahedral Voids
A tetrahedral void is a space surrounded by 4 atoms at the corners of a regular tetrahedron.
- Number per unit cell: 12 (2 per atom × 6 atoms)
- Radius: rt = 0.225R
- Accommodates: Small interstitial atoms like hydrogen and carbon
(B) Octahedral Voids
An octahedral void is surrounded by 6 atoms — 3 from the layer above and 3 from the layer below.
- Number per unit cell: 6 (1 per atom × 6 atoms)
- Radius: ro = 0.414R
- Accommodates: Larger interstitial atoms
These voids are of enormous practical importance. When carbon is dissolved in iron to make steel, the carbon atoms occupy the octahedral voids. Similarly, hydrogen storage in titanium-based materials involves hydrogen atoms sitting in the tetrahedral voids of the HCP lattice.
5. The c/a Ratio in Real Metals — Deviations from Ideal
In real metals, the c/a ratio deviates from the ideal value of 1.633 because atoms interact through quantum mechanical forces. Some metals are 'more elongated' (c/a > 1.633) and some are 'flattened' (c/a < 1.633) along the c-axis. These deviations significantly affect mechanical properties.
| Metal | Atomic Number | c/a Ratio | Deviation from Ideal | Primary Application |
|---|---|---|---|---|
| Cadmium (Cd) | 48 | 1.886 | +15.5% (most elongated) | Batteries, electroplating |
| Zinc (Zn) | 30 | 1.856 | +13.7% | Galvanising steel |
| Magnesium (Mg) | 12 | 1.624 | −0.5% (near ideal) | Lightweight structures |
| Cobalt (Co) | 27 | 1.623 | −0.6% | Magnetic materials, superalloys |
| Zirconium (Zr) | 40 | 1.593 | −2.4% | Nuclear reactor cladding |
| Titanium (Ti) | 22 | 1.587 | −2.8% | Aerospace, biomedical |
| Beryllium (Be) | 4 | 1.568 | −4.0% (most compressed) | Aerospace, nuclear |
| Ideal HCP | — | 1.633 | — | Theoretical reference |
- c/a > 1.633: The hexagonal prism is taller than ideal. Atoms are squeezed in the basal plane. Examples: Zn, Cd.
- c/a < 1.633: The prism is shorter and wider. Atoms are compressed along the c-axis. Examples: Ti, Be, Zr.
- c/a ≈ 1.633: Near-ideal packing. Examples: Mg, Co.
- Engineering consequence: Metals with c/a far from 1.633 often have fewer active slip systems, making them more brittle at room temperature.
6. Slip Systems in HCP — Why HCP Metals Are Less Ductile
6.1 What is a Slip System?
When a metal deforms plastically, the mechanism is slip — the sliding of one plane of atoms over an adjacent plane. Slip occurs most readily on the most densely packed planes and in the most densely packed directions.
A slip system is defined as a combination of one specific slip plane and one specific slip direction within that plane. The more slip systems a crystal has, the more ductile it is.
6.2 Slip Systems in HCP
In HCP, the close-packed basal plane — denoted (0001) — is the primary slip plane. Within this plane, there are 3 close-packed directions, giving:
| Crystal Structure | Primary Slip Plane(s) | Slip Directions / Plane | Slip Systems | Room-Temp. Ductility |
|---|---|---|---|---|
| BCC | {110} | 2 | 12 (primary) | Moderate |
| FCC | {111} | 3 | 12 | High |
| HCP | {0001} (basal) | 3 | 3 (basal only) | Low (at room temp.) |
With only 3 basal slip systems, an HCP crystal has fewer than the minimum 5 independent slip systems required (by the von Mises criterion) for general plastic deformation. However, at elevated temperatures, additional non-basal slip systems (prismatic {10‑10} and pyramidal {10‑11}) become activated, making HCP metals much more formable. This is why magnesium alloys are often processed at elevated temperatures.
7. HCP vs. FCC: A Detailed Structural Comparison
| Property | HCP | FCC |
|---|---|---|
| Stacking sequence | ABABAB··· | ABCABC··· |
| Stacking periodicity | 2 layers | 3 layers |
| Atoms per unit cell | 6 | 4 |
| Coordination number | 12 | 12 |
| Atomic packing factor | 74% (0.7405) | 74% (0.7405) |
| Lattice parameters | a and c (two needed) | a only (one needed) |
| Unit cell shape | Hexagonal prism | Cube with face atoms |
| Close-packed plane | (0001) basal | {111} planes |
| No. of close-packed planes | 1 | 4 |
| Primary slip systems | 3 (basal) | 12 |
| Room-temp. ductility | Low to moderate | High |
| Voids per atom | 1 oct + 2 tet | 1 oct + 2 tet |
| Example metals | Mg, Ti, Zn, Co, Zr | Al, Cu, Au, Ni, Pb |
HCP and FCC both pack atoms at 74% efficiency with 12-fold coordination. The difference is purely in symmetry and stacking sequence. This seemingly small structural difference leads to dramatically different mechanical behaviour (3 vs. 12 slip systems), different physical properties, and different alloy designs. It is a beautiful example of how atomic-scale structure controls macroscopic engineering properties.
8. Real Metals with HCP Structure and Their Applications
8.1 Magnesium (Mg) — The Lightest Structural Metal
Magnesium is the lightest engineering metal in practical use, with a density of only 1.74 g/cm³ — two-thirds the density of aluminium and one-quarter that of steel. It adopts the HCP structure at room temperature with a near-ideal c/a ratio of 1.624. Magnesium alloys are extensively used in automotive, aerospace, and portable electronics industries where weight reduction is critical.
8.2 Titanium (Ti) — The Aerospace Metal
Titanium exhibits allotropism: it is HCP (alpha phase) below 882°C and transforms to BCC (beta phase) above this temperature. Titanium alloys such as Ti-6Al-4V are used in aircraft frames, jet engine components, medical implants, and submarine hulls, owing to the high strength, excellent corrosion resistance, and good biocompatibility of the HCP phase.
8.3 Zinc (Zn) — The Corrosion Protector
Zinc has a significantly non-ideal c/a ratio of 1.856. It is most famously used for galvanising steel, where a thin zinc coating provides sacrificial cathodic protection to prevent rusting. Zinc is also a key component of brass (Zn-Cu alloy) and die-casting alloys.
8.4 Zirconium (Zr) — The Nuclear Metal
Zirconium has an extremely low neutron absorption cross-section, making it virtually transparent to neutrons. Zirconium alloys (particularly Zircaloy) are used as cladding tubes for nuclear fuel rods in water-cooled reactors.
8.5 Cobalt (Co) — The Magnetic HCP Metal
Cobalt is HCP below 417°C and FCC above this temperature. It is used in permanent magnets, magnetic recording media, high-temperature superalloys, and as a binder in cemented carbide cutting tools (WC-Co).
9. Describing Planes and Directions in HCP: Miller-Bravais Indices
In hexagonal crystals, the standard three-index Miller notation is insufficient because it does not reflect hexagonal symmetry. For this reason, hexagonal crystals use a four-index system called Miller-Bravais indices: (h k i l), where i = −(h+k).
9.1 The Four Axes of the Hexagonal System
- a₁: One of three equal in-plane axes, separated by 120° from each other.
- a₂: Second in-plane axis, 120° from a₁.
- a₃: Third in-plane axis (redundant but needed for symmetry), equal to −(a₁+a₂). This gives i = −(h+k).
- c: The vertical axis, perpendicular to the basal plane.
9.2 Important Planes in HCP
| Plane Name | Miller-Bravais Indices | Description |
|---|---|---|
| Basal plane | (0001) | The hexagonal base — the close-packed plane. Primary slip plane in HCP. |
| Prismatic plane | (10‑10) | Vertical planes parallel to the c-axis. Second slip plane at elevated temperatures. |
| Pyramidal plane | (10‑11) | Inclined planes. Active at high temperatures as additional slip planes. |
| Second-order pyramidal | (11‑22) | Steeper pyramidal planes. Important for <c+a> dislocation slip. |
9.3 The Close-Packed Direction in HCP
The most densely packed direction is the <11‑20> direction — connecting nearest neighbours within the basal plane. The Burgers vector of basal dislocations in HCP is b = (1/3)<11‑20>.
10. Solved Problems
Question: Magnesium has an HCP structure with a = 0.3209 nm and c = 0.5211 nm. Calculate the atomic radius of Mg.
Solution: In HCP, atoms touch along the in-plane edge a. Therefore a = 2R.
The tabulated atomic radius of Mg is 160 pm. Our calculated value is in excellent agreement. ✔
Question: Using a = 0.3209 nm, c = 0.5211 nm, calculate the c/a ratio and compare with the ideal value of 1.633.
The ideal value is 1.633. Magnesium's c/a = 1.624, which is only 0.5% below ideal. Magnesium is one of the closest to ideal HCP packing among all common metals. ✔
Question: Calculate the volume of the HCP unit cell of Magnesium (a = 0.3209 nm, c = 0.5211 nm).
Question: Calculate the theoretical density of Magnesium. (Atomic mass = 24.31 g/mol; NA = 6.022 × 10²³ atoms/mol)
The experimentally measured density of Mg is 1.738 g/cm³. Our calculation matches exactly. ✔
11. Practice Multiple Choice Questions
- (a) ABCABC
- (b) ABABAB ✔ Correct
- (c) AABBCC
- (d) ABACAB
- (a) 6
- (b) 8
- (c) 10
- (d) 12 ✔ Correct
- (a) 1.414
- (b) 1.732
- (c) 1.633 ✔ Correct
- (d) 1.500
- (a) 2
- (b) 4
- (c) 6 ✔ Correct
- (d) 8
- (a) 52%
- (b) 68%
- (c) 74% ✔ Correct
- (d) 91%
- (a) Aluminium (FCC)
- (b) Iron at room temperature (BCC)
- (c) Magnesium ✔ Correct — HCP
- (d) Copper (FCC)
- (a) 12 slip systems
- (b) Higher atomic packing factor than FCC
- (c) Only 3 primary basal slip systems ✔ Correct
- (d) Lower coordination number than BCC
- (a) Magnesium (1.624)
- (b) Titanium (1.587)
- (c) Cadmium (1.886) ✔ Correct
- (d) Beryllium (1.568)
12. Key Takeaways — What You Have Learned
- DEFINITION: HCP is formed by stacking close-packed hexagonal layers in an A-B-A-B sequence, where every other layer is directly above the first.
- UNIT CELL: A regular hexagonal prism with lattice parameters a and c. It is the only common crystal structure that requires two independent parameters.
- ATOM COUNT: 6 atoms per unit cell (12 corners × 1/6 + 2 faces × 1/2 + 3 interior = 2 + 1 + 3 = 6).
- COORDINATION NUMBER: 12 (6 in-plane + 3 above + 3 below). Maximum possible for identical spheres.
- IDEAL c/a RATIO: 1.633 (derived geometrically as 2√(2/3)). Real metals deviate because atoms are not perfect spheres.
- ATOMIC PACKING FACTOR: 74% — the theoretical maximum, shared with FCC.
- VOIDS: Each unit cell has 6 octahedral voids (r = 0.414R) and 12 tetrahedral voids (r = 0.225R).
- SLIP SYSTEMS: Only 3 primary basal slip systems, compared to 12 in FCC. This limits room-temperature ductility.
- HCP vs FCC: Both have APF = 74% and CN = 12, but differ in stacking (ABABAB vs ABCABC), slip systems (3 vs 12), and unit cell symmetry.
- KEY METALS: Magnesium (lightweight structures), Titanium (aerospace, biomedical), Zinc (galvanising), Zirconium (nuclear), Cobalt (magnets, superalloys).
This tutorial is part of the Crystal Structure Master Hub at AdvanceMaterialsLab.com
Next in this series: Bravais Lattices | Miller Indices | X-Ray Diffraction & Bragg's Law
© 2026 AdvanceMaterialsLab.com — All rights reserved
Dr. Rolly Verma
Explore More from Advance Materials Lab
Continue expanding your understanding of material behavior through our specialized resources on ferroelectrics and phase transitions. Each guide is designed for research scholars and advanced learners seeking clarity, precision, and depth in the field of materials science and nanotechnology.
🔗 Recommended Reads and Resources:
Ferroelectrics Tutorials and Research Guides — Comprehensive tutorials covering polarization, hysteresis, and ferroelectric device characterization.
Workshops on Ferroelectrics (2025–2027) — Upcoming training sessions and research-oriented workshops for hands-on learning.
Glossary — Ferroelectrics and Phase Transitions — Concise explanations of key terminologies to support your study and research work.
If you notice any inaccuracies or have constructive suggestions to improve the content, I warmly welcome your feedback. It helps maintain the quality and clarity of this educational resource. You can reach me at: advancematerialslab27@gmail.com
