Lattice and Basis in Crystallography
The Mathematical Foundation for Describing All Crystal Structures By Dr. Rolly Verma | AdvanceMaterialsLab.com | March 2026 | B.Sc. / M.Sc. Materials Science & EngineeringSeries: Crystal Structure Hub | Lecture: 02 of 10 | Prerequisite: Lecture 01 — Introduction to Crystal Structure
Reading time: 30 minutes | Includes: Lattice definition, basis concept, primitive vectors, mathematical framework, worked examples, real crystal structures, MCQs, key takeaways
SEO Keywords: lattice and basis crystallography, what is a lattice in materials science, crystal structure lattice plus basis, primitive lattice vectors, Bravais lattice, lattice basis crystal structure, crystallography tutorial
1. Connecting to Lecture 01
Welcome back to the Crystal Structure Hub. In Lecture 01, we established the most important principle in materials science: the properties of a material are determined not only by its chemical composition, but fundamentally by how its atoms are arranged in space. We saw this through the dramatic contrast between diamond and graphite — identical atoms, completely different crystal arrangements, completely different properties.
We also encountered the idea of periodicity — the regular, repeating nature of atomic arrangements in crystalline solids. But to work with crystal structures rigorously — to calculate their properties, predict their behaviour, and design new materials — we need a precise mathematical language to describe this periodicity. That language is built on two foundational concepts: the lattice and the basis.
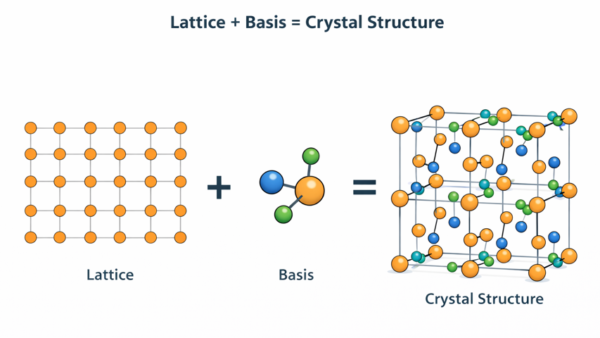
Crystal Structure = Lattice + Basis
By the end of this lecture, you will understand exactly what each term in this equation means, why this decomposition is so powerful, and how it applies to real materials ranging from simple metals to complex ceramics and quantum materials.
2. What is a Lattice?
2.1 Building Intuition — The Wallpaper Analogy
Before we introduce the formal definition, let us build intuition through a familiar example. Imagine a wallpaper with a repeating floral pattern. Look carefully at any single flower in the pattern, and then look at any other flower. They are in exactly the same position relative to their surroundings — the same distance from their nearest neighbours, the same orientation in space. If you closed your eyes, moved to another flower, and opened them again, you would not be able to tell you had moved.
Now imagine reducing that wallpaper to its simplest repeating framework — not the flowers themselves, but just a dot at the centre of each flower. You would have a regular grid of equally spaced dots. That grid is the lattice. It captures the geometry of repetition without specifying what is being repeated.
Think of a tiled floor in a large room. Each tile is identical and placed at regular intervals. The pattern of tile centres forms a perfectly regular grid — a lattice. The design painted on each tile (a flower, a geometric pattern, a colour gradient) is the basis. The complete floor = the grid (lattice) + the tile design (basis).
This is exactly how crystallographers describe crystal structures. The lattice is the grid of repeating positions; the basis is the group of atoms placed at each position.
2.2 Formal Definition of a Lattice
With that intuition established, we can now state the formal definition that is used throughout solid-state physics and materials science, as given in the authoritative textbook by Kittel — Introduction to Solid State Physics:
A lattice is an infinite array of points in space arranged such that the environment of every point is identical to that of every other point.
Every lattice point has exactly the same neighbourhood — the same number of nearest neighbours, at the same distances, in the same directions. This perfect translational equivalence is what makes a lattice a lattice.
Three points from this definition deserve careful attention:
- A lattice is a mathematical abstraction, not a physical object. It is a set of points in space — a geometric framework. It does not, by itself, represent atoms. Atoms are introduced through the basis, which we will define shortly.
- Every lattice point has an identical environment. This is the defining constraint. If even one point has a different environment (different number of neighbours, different distances), it is not a perfect lattice.
- The lattice extends infinitely in all directions. Real crystals are finite, but they contain so many repeating units (typically ~10²³ atoms per mole) that the approximation of an infinite lattice is extraordinarily accurate for describing bulk properties.
2.3 Translation Symmetry — The Defining Property
The property that makes a lattice mathematically special is translation symmetry. If you take the entire lattice and shift it by the vector connecting any two lattice points, the lattice looks exactly the same as before. Every point coincides with another point.
This is not merely a mathematical curiosity — it has profound physical consequences. Because a crystal lattice has translation symmetry, electrons moving through the crystal experience a repeating potential. This repetition is precisely what gives rise to the electronic band structure, as we discussed in Lecture 01. Bloch's theorem — the foundational result of solid-state electronic structure theory — applies specifically and exclusively to systems with translation symmetry. Without the lattice, there is no band theory; without band theory, there is no semiconductor physics.
3. Mathematical Description — Primitive Lattice Vectors
3.1 Primitive Lattice Vectors
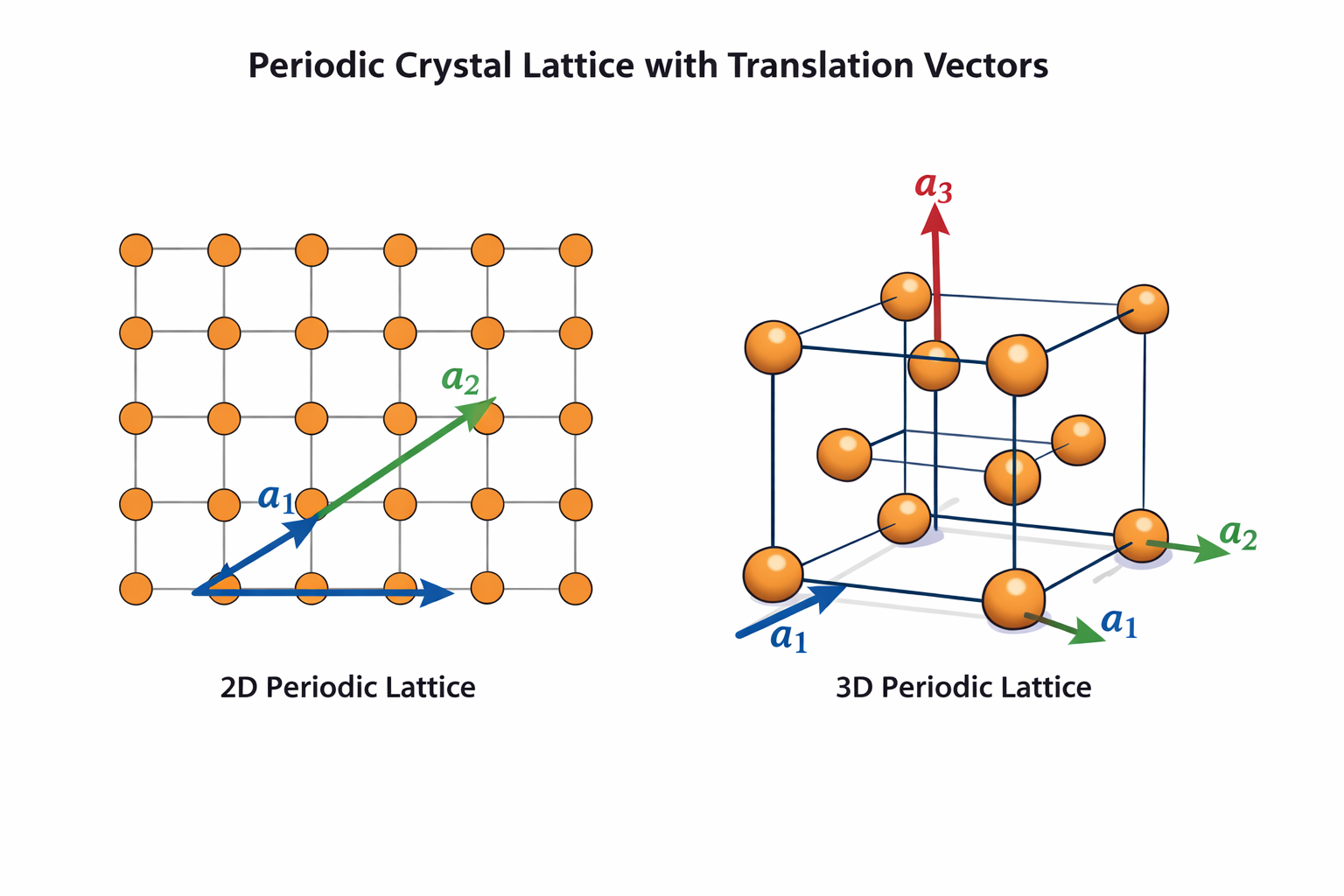
To work with a lattice quantitatively, we need a way to generate all lattice points mathematically. This is done using primitive lattice vectors (also called primitive translation vectors), denoted in three-dimensional space as:
a₁, a₂, a₃
These three vectors define the shortest translation distances along the three independent directions in which the lattice repeats. They are the fundamental building blocks of the lattice geometry. Their magnitudes |a₁|, |a₂|, |a₃| are called the lattice parameters (commonly denoted a, b, c), and the angles between them are denoted α (between a₂ and a₃), β (between a₁ and a₃), and γ (between a₁ and a₂). Together, these six quantities (a, b, c, α, β, γ) completely specify the geometry of any crystal lattice — this is covered in full detail in our Lecture 03 on Unit Cell and Lattice Parameters.
The primitive lattice vectors are the smallest vectors that can generate the entire lattice by integer combinations. There is no smaller set of vectors from which every lattice point can be reached. In a simple cubic lattice, for example, the three primitive vectors are simply three equal vectors pointing along the x, y, and z axes: a₁ = ax̂, a₂ = aŷ, a₃ = aẑ. In more complex lattices, they may point in non-perpendicular directions and have different magnitudes.
3.2 The Lattice Vector Equation
Using the three primitive vectors, the position of any lattice point in the crystal can be expressed as a lattice vector R:
R = n₁a₁ + n₂a₂ + n₃a₃
Here, n₁, n₂, n₃ are integers (positive, negative, or zero). This single equation is remarkably powerful: by substituting every possible combination of integer values for n₁, n₂, n₃, you generate the entire infinite lattice — every single lattice point, in any crystal system. The lattice is fully and completely described by just three vectors.
This equation tells us something elegant: the entire infinite, periodic structure of a crystal can be generated from three vectors and a set of integers. This mathematical compactness is what makes the lattice formalism so powerful for calculations in solid-state physics and crystallography.
4. What is a Basis?
We have established that a lattice is a mathematical framework of repeating points. But the lattice alone describes no real material — it is just geometry. To describe an actual crystal, we must specify what exists at each lattice point. That is the role of the basis.
The basis is a group of one or more atoms (with specified positions relative to the lattice point) that is attached to every lattice point. The same basis is placed at every lattice point in an identical orientation. When the basis repeats with the periodicity of the lattice, the complete crystal structure is generated.
Several important points follow from this definition:
- A basis can be a single atom — as in many elemental metals. In this case, one metal atom sits at each lattice point.
- A basis can be two or more atoms of the same element — for example, in the diamond cubic structure, the basis consists of two carbon atoms, even though the material contains only one element.
- A basis can contain atoms of different elements — as in all compound crystal structures (NaCl, BaTiO₃, Si). In NaCl, the basis is one Na atom and one Cl atom at specific positions relative to each FCC lattice point.
- The position of each atom in the basis is expressed as a vector rᵢ measured from the lattice point, usually written in terms of the primitive vectors: rᵢ = ua₁ + va₂ + wa₃, where u, v, w are fractional numbers between 0 and 1.
4.1 The Basis in Real Crystals — Three Levels of Complexity
The elegance of the lattice-plus-basis framework becomes clear when we apply it to real materials across different levels of complexity:
| Material | Lattice Type | Basis | Atoms per Basis |
|---|---|---|---|
| Copper (Cu) | Face-Centred Cubic (FCC) | 1 Cu atom at (0,0,0) | 1 |
| Iron (α-Fe) | Body-Centred Cubic (BCC) | 1 Fe atom at (0,0,0) | 1 |
| Sodium chloride (NaCl) | FCC | Na at (0,0,0) + Cl at (½,0,0) | 2 |
| Diamond (C) / Silicon (Si) | FCC | C at (0,0,0) + C at (¼,¼,¼) | 2 |
| Hexagonal close-packed (HCP) | Simple Hexagonal | Atom at (0,0,0) + atom at (⅓,⅔,½) | 2 |
| Barium titanate (BaTiO₃) | Simple Cubic | Ba at (0,0,0), Ti at (½,½,½), 3×O at (½,½,0) etc. | 5 |
| Fullerene (C₆₀) | FCC | 60 C atoms per basis in icosahedral arrangement | 60 |
This table reveals something remarkable: many structurally different materials — copper, sodium chloride, diamond, and silicon — all share the same FCC lattice. What distinguishes them is their basis. The lattice captures the geometry of repetition; the basis captures the chemistry and structural detail.
5. Crystal Structure = Lattice + Basis
We are now in a position to fully appreciate the fundamental equation of crystallography. With the lattice vector R locating each lattice point and the basis position rᵢ locating each atom within the basis, the position of the ith atom in the crystal is:
Position of atom i = R + rᵢ
= (n₁a₁ + n₂a₂ + n₃a₃) + rᵢ
By letting n₁, n₂, n₃ range over all integers and i range over all atoms in the basis, this expression generates the position of every atom in the entire crystal. The entire structure — no matter how complex — is encoded in three primitive vectors and a list of basis positions.
- Universality: Every crystal structure that exists — from the simplest metal to the most complex protein crystal with thousands of atoms per unit cell — can be described using exactly this framework.
- Separation of concerns: The lattice captures periodicity and geometry; the basis captures chemistry and local structure. These can be studied and modified independently.
- Computational efficiency: Instead of specifying the positions of 10²³ atoms, you specify three primitive vectors and a few basis positions. Computer simulations of crystals rely entirely on this compactness.
- Predictive power: Because all physically measurable properties (diffraction patterns, band structures, phonon spectra) depend on the periodic structure, knowing lattice + basis is sufficient to calculate virtually any property of the crystal.
6. Bravais Lattices — The Complete Classification
A natural question arises: how many distinct lattice geometries are possible in three-dimensional space? The answer was determined by the French mathematician Auguste Bravais in 1848, and it is a finite number: exactly 14. These are the 14 Bravais lattices, and they represent all possible ways that points can be periodically arranged in three-dimensional space while maintaining translational symmetry, as formally established by the International Union of Crystallography.
| Crystal System | Bravais Lattices | Lattice Parameters | Example Material |
|---|---|---|---|
| Cubic | Simple (SC), BCC, FCC | a = b = c, α = β = γ = 90° | Fe (BCC), Cu (FCC), Po (SC) |
| Tetragonal | Simple, Body-centred | a = b ≠ c, α = β = γ = 90° | Indium, MnF₂ |
| Orthorhombic | Simple, Base-centred, Body-centred, Face-centred | a ≠ b ≠ c, α = β = γ = 90° | Sulphur, gallium |
| Hexagonal | Simple | a = b ≠ c, α = β = 90°, γ = 120° | Mg (HCP), Zn, graphite |
| Rhombohedral | Simple | a = b = c, α = β = γ ≠ 90° | Bismuth, calcite |
| Monoclinic | Simple, Base-centred | a ≠ b ≠ c, α = γ = 90°, β ≠ 90° | Gypsum, orthoclase |
| Triclinic | Simple | a ≠ b ≠ c, α ≠ β ≠ γ ≠ 90° | Kaolinite, turquoise |
The three cubic Bravais lattices — Simple Cubic (SC), Body-Centred Cubic (BCC), and Face-Centred Cubic (FCC) — are the most commonly encountered in introductory materials science because the majority of elemental metals adopt one of these three structures. The HCP structure is built on a simple hexagonal Bravais lattice with a two-atom basis.
The fact that there are exactly 14 Bravais lattices — no more, no fewer — is a mathematical theorem proved by considering all possible combinations of symmetry operations (translation, rotation, reflection, inversion) that leave a lattice invariant. It is not an empirical observation but a rigorous mathematical result. Similarly, the fact that there are exactly 230 space groups (the complete set of ways that symmetry operations can combine in three dimensions) is a theorem of group theory, as catalogued in the authoritative International Tables for Crystallography (IUCr).
7. Worked Examples — Real Crystal Structures
Lattice: Face-Centred Cubic (FCC). Primitive vectors point from the corner of the cube to the three nearest face centres. Lattice parameter a = 0.3615 nm.
Basis: One Cu atom at position (0, 0, 0) relative to each FCC lattice point.
Crystal structure of Cu = FCC lattice + basis of 1 Cu atom at (0,0,0)
Since there is one atom per basis, and the FCC unit cell contains 4 lattice points equivalent, there are 4 Cu atoms per conventional unit cell. This agrees with the standard result of 4 atoms per FCC unit cell.
Lattice: Face-Centred Cubic (FCC). Lattice parameter a = 0.5640 nm.
Basis: Two atoms — Na and Cl — at specific positions relative to each FCC lattice point:
Na at fractional coordinates: (0, 0, 0)
Cl at fractional coordinates: (½, 0, 0)
i.e. Cl displaced by a/2 along the x-axis from Na
The crystal structure of NaCl is therefore: the FCC lattice with this two-atom basis repeated at every lattice point. This produces the characteristic rock salt structure — alternating Na⁺ and Cl⁻ ions, each with 6 nearest neighbours of the opposite type (coordination number = 6). The conventional unit cell contains 4 formula units of NaCl = 4 Na + 4 Cl = 8 atoms total.
Lattice: Face-Centred Cubic (FCC).
Basis: Two atoms of the same element at positions:
Atom 1 at: (0, 0, 0)
Atom 2 at: (¼, ¼, ¼)
The second atom sits at one-quarter of the way along the body diagonal from the first. This two-atom basis on an FCC lattice generates the diamond cubic structure — a tetrahedral network where each atom has 4 nearest neighbours. This structure is the same for both diamond (C, a = 0.357 nm) and silicon (Si, a = 0.543 nm). The lattice type and basis description are identical; only the lattice parameter differs due to the different atomic radii.
8. Key Properties of a Lattice
Now that we understand what a lattice is and how it combines with a basis, let us consolidate the key mathematical properties that make the lattice concept so useful in crystallography and solid-state physics.
8.1 Translational Symmetry
If the entire lattice is shifted by any lattice vector R = n₁a₁ + n₂a₂ + n₃a₃, the resulting configuration is identical to the original. This is the defining mathematical property of a lattice, and it directly underlies Bloch's theorem in quantum mechanics — the foundation for all band structure calculations of crystalline materials.
8.2 All Lattice Points Are Equivalent
No lattice point is special or unique. Every point has exactly the same environment — the same number of nearest neighbours, at the same distances, in the same relative directions. This equivalence is what allows the same basis to be repeated identically at every lattice point.
8.3 The Lattice Is a Mathematical Abstraction
This cannot be emphasised enough: the lattice does not represent atoms directly. It represents positions in space. The physical atoms — with their specific species, positions, and bonding — enter through the basis. Many students confuse "corner atoms" in a unit cell diagram with lattice points; it is important to remember that atoms sit at or near lattice points (via the basis), but they are not the lattice points themselves.
8.4 Primitive vs Conventional Cells
The smallest possible unit cell that captures all lattice information is the primitive cell — it contains exactly one lattice point and reproduces the lattice by translation alone. However, for cubic, tetragonal, and other high-symmetry lattices, a larger conventional cell is often used because it more clearly displays the crystal symmetry, even though it contains more than one lattice point. This distinction is discussed in full in our Lecture 03 on Unit Cell and Lattice Parameters.
9. Applications in Materials Science
The lattice-and-basis framework is not merely a theoretical convenience — it is the working language of experimental and computational materials science across all subfields.
9.1 X-ray Diffraction and Structure Determination
When X-rays interact with a crystal, they scatter from every atom. The diffraction pattern — the set of observed intensities at different angles — depends on both the lattice geometry (which determines the angles at which constructive interference occurs, via Bragg's Law) and the basis (which determines the intensities of each diffraction peak through the structure factor). By analysing a complete diffraction pattern, crystallographers can reverse-engineer both the lattice type and the basis — determining the complete crystal structure. This is the primary tool of structural crystallography at the Cambridge Crystallographic Data Centre and every modern materials characterisation laboratory.
9.2 Electronic Band Structure Calculations
The electronic properties of a crystal — its band structure, density of states, and Fermi surface — are computed by solving the Schrödinger equation for electrons in the periodic potential created by the crystal's lattice and basis. The periodicity of the lattice is encoded in the reciprocal lattice (the Fourier transform of the real-space lattice), and the basis determines the strength of the periodic potential at each reciprocal lattice point through the structure factor. This underpins all density functional theory (DFT) calculations used to design new semiconductors, battery materials, and quantum devices.
9.3 Nanotechnology and Thin Film Engineering
When growing crystalline thin films — for example, depositing a magnetic film on a semiconductor substrate — the lattice mismatch between substrate and film creates strain that modifies both the film's crystal structure and its electronic, magnetic, and ferroelectric properties. The recently demonstrated topological crystalline insulator in bilayer SnTe (Nature Communications, 2025) is a direct example: the compressive strain imposed by the NbSe₂ substrate modifies the SnTe lattice parameters, stabilising a topological quantum phase that does not exist in the unstrained material. The basis (SnTe atom positions) remains the same; the lattice geometry changes under strain — and that geometric change transforms the quantum electronic state.
9.4 Semiconductor Doping and Defect Engineering
When impurity atoms are introduced into a semiconductor lattice (doping), they substitute for lattice basis atoms or occupy interstitial positions between lattice points. The difference in size, charge, and bonding between the impurity and the host basis atom creates local strain fields and introduces electronic states within the band gap. All of silicon's remarkable properties as a semiconductor — its ability to be engineered into n-type or p-type material — arise from understanding how substitutional impurities modify the crystal's lattice-plus-basis structure at specific sites.
9.5 Ferroelectric and Piezoelectric Materials
In ferroelectric perovskite materials like barium titanate (BaTiO₃), the basis positions of the constituent atoms within the unit cell determine whether the crystal is ferroelectric. Above the Curie temperature, Ti⁴⁺ sits at the exact centre of the oxygen octahedron — centrosymmetric basis. Below the Curie temperature, Ti⁴⁺ displaces off-centre — breaking the centrosymmetry of the basis — and spontaneous polarisation appears. The lattice remains cubic; only the basis changes. Yet this small displacement in the basis position generates the entire phenomenon of ferroelectricity, as demonstrated in detail in Dr. Verma's own research on perovskite crystal structures.
10. Series Navigation
11. Practice Questions
- (a) A lattice contains atoms; a crystal structure contains only geometric points
- (b) A lattice is three-dimensional; a crystal structure can be one-dimensional
- (c) A lattice is an abstract geometric framework of points; a crystal structure is the lattice with a basis of atoms attached ✔
- (d) A crystal structure contains only one type of atom; a lattice can contain multiple types
- (a) (0, 0, 0) ✔ — all coefficients zero gives R = 0
- (b) (1, 1, 1)
- (c) (½, ½, ½)
- (d) Any set of equal integers
- (a) The Bravais lattice type
- (b) The number of atoms in the basis
- (c) The fractional coordinates of the basis atoms
- (d) The lattice parameter a (the actual physical size of the unit cell) ✔
- (a) 7 (one for each crystal system)
- (b) 32 (one for each point group)
- (c) 14 ✔ — proved mathematically by Auguste Bravais in 1848
- (d) 230 (one for each space group)
- (a) 2 atoms (1 Na + 1 Cl)
- (b) 4 atoms
- (c) 8 atoms ✔ — 4 lattice points × 2 atoms per basis = 8 atoms (4 Na + 4 Cl)
- (d) 6 atoms
- (a) The Bravais lattice type changes from cubic to hexagonal
- (b) The number of atoms in the basis changes
- (c) The fractional coordinates of the basis (positions of atoms within the unit cell) change ✔
- (d) The primitive lattice vectors change direction
12. Key Takeaways
- THE FUNDAMENTAL EQUATION: Crystal Structure = Lattice + Basis. Every crystal structure, no matter how complex, can be described by this decomposition.
- THE LATTICE: An infinite array of points in space where every point has an identical environment. The lattice is a mathematical abstraction — it does not represent atoms directly. It represents geometric positions where atoms can be placed.
- TRANSLATION SYMMETRY: The defining property of a lattice — shifting it by any lattice vector leaves it unchanged. This property underlies Bloch's theorem and all electronic band structure calculations.
- PRIMITIVE VECTORS: Three independent vectors a₁, a₂, a₃ that generate all lattice points via R = n₁a₁ + n₂a₂ + n₃a₃ (integers n₁, n₂, n₃). Three vectors and a set of integers describe the entire infinite lattice.
- THE BASIS: A group of one or more atoms at specified positions relative to each lattice point. The basis introduces the actual chemistry — which atoms, where exactly. The same basis is attached at every lattice point.
- ATOMIC POSITIONS: Position of any atom in the crystal = R + rᵢ, where R is the lattice vector and rᵢ is the atom's position within the basis.
- 14 BRAVAIS LATTICES: All possible distinct periodic point arrangements in 3D reduce to exactly 14 Bravais lattices — a mathematical theorem proved by Auguste Bravais in 1848.
- SAME LATTICE, DIFFERENT BASIS: Copper, NaCl, diamond, and silicon all use the FCC lattice but have different bases — showing how the lattice and basis independently contribute to the final structure.
- APPLICATIONS: The lattice-basis framework is the working language of X-ray diffraction analysis, band structure calculations, thin film engineering, semiconductor doping, and ferroelectric materials design.
- COMING NEXT: Lecture 03 will define the unit cell and lattice parameters (a, b, c, α, β, γ) — the six numbers that quantitatively describe the geometry of any crystal lattice.
References
All references are in IEEE citation style. All sources are peer-reviewed journals, internationally recognised textbooks, or authoritative academic databases.
- C. Kittel, Introduction to Solid State Physics, 8th ed. Hoboken, NJ, USA: John Wiley & Sons, 2005, ch. 1, pp. 1–26. [Wiley] — Primary reference for the formal definition of lattice, primitive vectors, and the lattice vector equation.
- N. W. Ashcroft and N. D. Mermin, Solid State Physics. Philadelphia, PA, USA: Holt, Rinehart and Winston, 1976, ch. 4–5. — Classic graduate reference for Bravais lattices, the reciprocal lattice, and the mathematical foundations of crystal periodicity.
- W. D. Callister Jr. and D. G. Rethwisch, Materials Science and Engineering: An Introduction, 10th ed. Hoboken, NJ, USA: John Wiley & Sons, 2018, ch. 3. [Wiley] — Standard undergraduate reference for crystal structures, Bravais lattices, and the lattice-basis framework applied to common engineering materials.
- A. Kelly and K. M. Knowles, Crystallography and Crystal Defects, 2nd ed. Chichester, UK: John Wiley & Sons, 2012. [Wiley] — Comprehensive reference for crystallographic concepts including primitive vectors, basis description, and crystal symmetry in the context of materials defects.
- International Union of Crystallography (IUCr), "Teaching Pamphlets: Crystal Symmetry and Space Groups," IUCr Education Pamphlets, no. 9. Chester, UK: IUCr, 2002. [iucr.org — full text] — Authoritative reference for the 14 Bravais lattices, 7 crystal systems, and the mathematical classification of lattice symmetry.
- IUCr, International Tables for Crystallography, Volume A: Space-Group Symmetry, 6th ed. Chester, UK: IUCr, 2016. [IUCr International Tables] — The definitive reference for all 230 space groups and complete crystallographic symmetry data for all known crystal structures.
- A. Jain et al., "Commentary: The Materials Project: A materials genome approach to accelerating materials innovation," APL Mater., vol. 1, no. 1, Art. no. 011002, Jul. 2013, doi: 10.1063/1.4812323. [Materials Project Database] — Open-access database used for verifying lattice parameters and crystal structure data for all worked examples (Cu, NaCl, Si, BaTiO₃).
- L. Jing et al., "Strain-induced two-dimensional topological crystalline insulator in bilayer SnTe," Nature Communications, vol. 16, Art. no. 6304, Jul. 2025, doi: 10.1038/s41467-025-67520-y. [Nature Communications — Open Access] — 2025 experimental example demonstrating how strain-induced changes to the crystal lattice stabilise new quantum phases — referenced in Section 9.3.
- R. Verma and S. K. Rout, "Frequency-dependent ferro–antiferro phase transition and internal bias field influenced piezoelectric response of donor and acceptor doped bismuth sodium titanate ceramics," J. Appl. Phys., vol. 126, no. 9, Art. no. 094103, Sep. 2019, doi: 10.1063/1.5111505. [AIP — DOI: 10.1063/1.5111505] — Author's peer-reviewed research directly relevant to Section 9.5 — demonstrating how basis position changes in BNT perovskites govern ferroelectric and piezoelectric behaviour.
- R. Verma and S. K. Rout, "The Mystery of Dimensional Effects in Ferroelectricity," in Recent Advances in Multifunctional Perovskite Materials, London, UK: IntechOpen, 2022, doi: 10.5772/intechopen.104435. [IntechOpen — Open Access] — Author's chapter on crystal structure-property relationships in perovskite materials, directly relevant to the BaTiO₃ discussion in Section 9.5.
This is Lecture 02 of the Crystal Structure Hub Series at AdvanceMaterialsLab.com
Next Lecture: Unit Cell and Lattice Parameters →
© 2026 AdvanceMaterialsLab.com — Dr. Rolly Verma | All rights reserved
Dr. Rolly Verma
If you notice any inaccuracies or have constructive suggestions to improve the content, I warmly welcome your feedback. It helps maintain the quality and clarity of this educational resource. You can reach me at: advancematerialslab27@gmail.com
Explore More from Advance Materials Lab
Continue expanding your understanding of material behavior through our specialized resources on ferroelectrics and phase transitions. Each guide is designed for research scholars and advanced learners seeking clarity, precision, and depth in the field of materials science and nanotechnology.
🔗 Recommended Reads and Resources:
Ferroelectrics Tutorials and Research Guides — Comprehensive tutorials covering polarization, hysteresis, and ferroelectric device characterization.
Workshops on Ferroelectrics (2025–2027) — Upcoming training sessions and research-oriented workshops for hands-on learning.
Glossary — Ferroelectrics and Phase Transitions — Concise explanations of key terminologies to support your study and research work.
