Crystal Structure Hub — Lecture 05
Packing Fraction and Theoretical Density in Crystal Structures
A Complete Derivation for SC, BCC, FCC & HCP — With Solved Problems & MCQs By Dr. Rolly Verma | AdvanceMaterialsLab.com | March 2026 | B.Sc. / M.Sc. Materials Science & EngineeringTopic: Packing Fraction & Theoretical Density | Level: Undergraduate / Postgraduate
Reading time: 35 minutes | Includes: Theory, Full Derivations for all 4 crystal structures, Tables, MCQs, Solved Problems
SEO Keywords: packing fraction crystal structure, atomic packing factor, APF BCC FCC HCP simple cubic, theoretical density calculation, crystal structure materials science, packing efficiency derivation
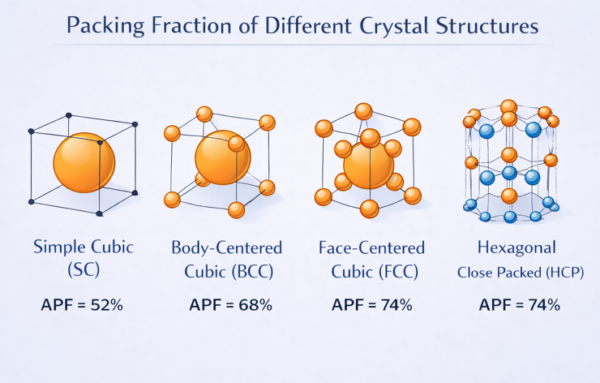
1. Introduction: Why Do Atoms Pack the Way They Do?
Welcome to this tutorial. In your study of materials science, you have already encountered the idea that atoms arrange themselves in regular, repeating patterns called crystal structures. Now, we take the next step and ask a question that is both practical and intellectually satisfying: how efficiently do those atoms fill the space available to them?
Imagine you are packing spherical oranges into a cardboard box. No matter how carefully you arrange them, there will always be gaps between the fruits — it is geometrically impossible to fill a box completely with spheres. Atoms in a crystal face exactly the same challenge. The fraction of space that is actually occupied by atoms — as opposed to the empty gaps between them — is called the Atomic Packing Factor (APF) or Packing Fraction.
This single number tells us an enormous amount about a material. It influences the density, the ease of plastic deformation, the diffusion of other atoms through the crystal, and even mechanical properties like hardness and ductility. Understanding how to calculate the packing fraction — and why it differs between crystal structures — is therefore one of the most foundational skills in materials science, as emphasised in the authoritative textbook by Callister and Rethwisch — Materials Science and Engineering.
- What atomic packing factor (APF) is and why it matters
- How to derive APF from first principles for all four major crystal structures: Simple Cubic (SC), Body-Centred Cubic (BCC), Face-Centred Cubic (FCC), and Hexagonal Close-Packed (HCP)
- How to calculate the theoretical density of any crystalline material from its crystal structure data
- How to compare and contrast the packing efficiency of all major structures
- How to solve numerical problems on density and APF
2. Core Concepts: Building the Foundation
2.1 What is Atomic Packing Factor (APF)?
The Atomic Packing Factor (APF) is defined as the fraction of the total volume of a unit cell that is actually occupied by atoms. It is a dimensionless quantity — a pure number between 0 and 1 — and is sometimes expressed as a percentage.
APF = Volume occupied by atoms in unit cell / Total volume of unit cell
Since atoms are modelled as hard spheres, the volume of each atom is (4/3)πR³, where R is the atomic radius. The total volume of the unit cell depends on the crystal structure — it is the volume of the cube for cubic systems (as classified by the International Union of Crystallography), or the hexagonal prism for HCP.
- An APF of 1.0 would mean 100% of the space is filled — this is impossible for spheres of uniform size.
- The maximum possible APF for identical spheres is 0.7405 (74.05%), achieved by both FCC and HCP structures.
- The remaining space (1 − APF) is the void fraction — empty space in the form of interstitial voids.
2.2 The Hard Sphere Model
Throughout our derivations, we treat each atom as a rigid, incompressible sphere of radius R — the hard sphere model. The key consequence: in a close-packed direction, neighbouring atoms are assumed to be touching — the centre-to-centre distance equals exactly 2R. This gives us the relationship between the lattice parameter a and the atomic radius R for each structure.
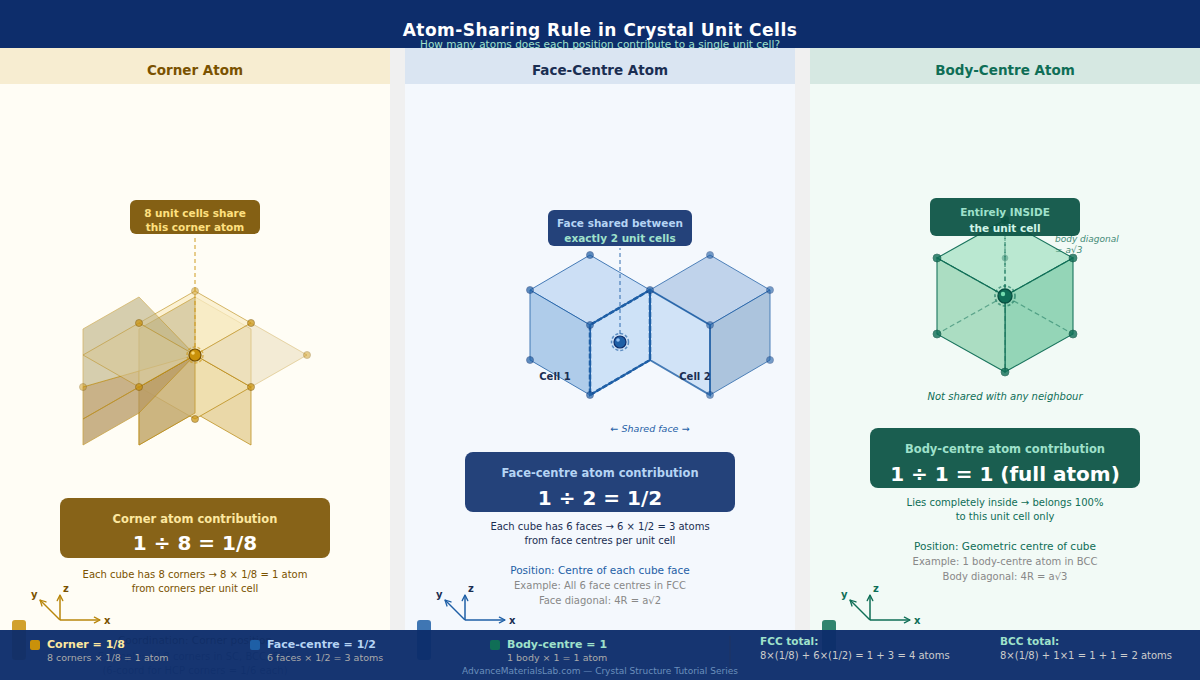
2.3 The Unit Cell — Our Counting Domain
When we calculate APF, we count atoms using the sharing rule — atoms on the boundary of a unit cell are shared with neighbouring cells:
- Corner atom: shared by 8 unit cells → contributes 1/8
- Edge atom: shared by 4 unit cells → contributes 1/4
- Face-centre atom: shared by 2 unit cells → contributes 1/2
- Body-centre atom: entirely inside → contributes 1
3. Simple Cubic (SC) Structure — The Simplest Case
3.1 Visualising the Simple Cubic Structure
The Simple Cubic (SC) structure is the most straightforward crystal structure — the ideal starting point. Atoms are placed only at the eight corners of a cube. There are no atoms on the faces, edges, or inside the cube. Looking at the structure from any direction, you see a perfectly square grid of atoms.
Think of stacking identical cubic cardboard boxes in a warehouse — one at each corner position, with no boxes filling the interior. This is the simple cubic structure: atoms at corners only, with a large empty interior. It is elegant but very wasteful of space.

3.2 Atoms per Unit Cell (SC)
N = 8 corners × (1/8) = 1 atom per unit cell
3.3 Relationship Between a and R (SC)
In SC, atoms touch along the cube edge. The edge length a equals the diameter of one atom:
a = 2R ⟹ R = a / 2
3.4 APF Derivation for Simple Cubic
Step 1: Volume of atoms in unit cell
V_atoms = 1 × (4/3)πR³ = (4/3)πR³
Step 2: Volume of unit cell
V_cell = a³ = (2R)³ = 8R³
Step 3: Calculate APF
APF = (4/3)πR³ / 8R³ = 4π / 24 = π/6
APF (SC) = π/6 = 0.5236 ≈ 52.36%
Only 52% of the available space is occupied — nearly half the volume is empty. This is why the SC structure is rare in nature. The only elemental metal adopting SC at standard conditions is Polonium (Po) — verified in the NIST Crystal Structure Database. Each atom has only 6 nearest neighbours (CN = 6).
3.5 Why Is SC Structure Rare?
A lower coordination number means fewer bonds per atom, which means lower binding energy and therefore a less stable structure. Nature prefers more tightly packed arrangements — structures with higher coordination numbers wherever possible.
4. Body-Centred Cubic (BCC) Structure
4.1 Visualising the BCC Structure
The BCC structure improves upon SC by placing one additional atom at the geometric centre of the cube — the body centre — in addition to the 8 corner atoms. This central atom nestles exactly in the middle, touching all 8 corner atoms simultaneously along the body diagonal.
Iron (Fe) below 912°C (alpha-iron) | Chromium (Cr) | Tungsten (W) | Molybdenum (Mo) | Vanadium (V) | Niobium (Nb) | Tantalum (Ta)
These metals tend to have high melting points and moderate ductility — directly related to the BCC structure, as documented in the ASM International Metals Handbook.

4.2 Atoms per Unit Cell (BCC)
N = 8 corners × (1/8) + 1 body centre × 1 = 1 + 1 = 2 atoms
4.3 Relationship Between a and R (BCC)
Atoms touch along the body diagonal (length = a√3):
4R = a√3 ⟹ R = (√3/4) × a or a = 4R / √3
4.4 APF Derivation for BCC
Step 1: Volume of atoms
V_atoms = 2 × (4/3)πR³ = (8/3)πR³
Step 2: Volume of unit cell
a = 4R/√3 ⟹ V_cell = a³ = (4R/√3)³ = 64R³ / (3√3)
Step 3: Calculate APF
APF = [(8/3)πR³] / [64R³ / (3√3)]
= [(8/3)π × (3√3)] / 64
= (8π√3) / 64 = π√3 / 8
APF (BCC) = π√3/8 = 0.6802 ≈ 68.02%
A significant improvement over SC (52%). The body-centre atom raises packing efficiency by 16 percentage points. For a complete structural analysis of BCC, see our dedicated tutorial on BCC Crystal Structure. Each atom has 8 nearest neighbours (CN = 8).
5. Face-Centred Cubic (FCC) Structure
5.1 Visualising the FCC Structure
The FCC structure places one atom at the centre of each of the six faces, in addition to the 8 corner atoms. This is much more densely packed than BCC and achieves the maximum theoretical packing efficiency.
Aluminium (Al) | Copper (Cu) | Gold (Au) | Silver (Ag) | Nickel (Ni) | Lead (Pb) | Platinum (Pt) | Iron (Fe) above 912°C
FCC metals are generally highly ductile and excellent electrical conductors — a direct consequence of 12-fold coordination and 12 abundant slip systems.

5.2 Atoms per Unit Cell (FCC)
N = 8 corners × (1/8) + 6 face centres × (1/2) = 1 + 3 = 4 atoms
5.3 Relationship Between a and R (FCC)
Atoms touch along the face diagonal (length = a√2):
4R = a√2 ⟹ R = a / (2√2) or a = 2√2 × R
5.4 APF Derivation for FCC
Step 1: Volume of atoms
V_atoms = 4 × (4/3)πR³ = (16/3)πR³
Step 2: Volume of unit cell
a = 2√2 R ⟹ V_cell = (2√2 R)³ = 16√2 R³
Step 3: Calculate APF
APF = [(16/3)πR³] / [16√2 R³]
= 16π / (3 × 16√2)
= π / (3√2)
APF (FCC) = π/(3√2) = 0.7405 ≈ 74.05%
FCC achieves the maximum theoretical packing efficiency. Every atom has 12 nearest neighbours (CN = 12) — the highest possible. The 12 available slip systems explain why FCC metals are the most ductile of all metallic structures. Explore our full FCC Crystal Structure tutorial for complete derivations.
6. Hexagonal Close-Packed (HCP) Structure
6.1 Visualising the HCP Structure
The HCP structure achieves the same maximum packing efficiency as FCC (74%) but through a completely different arrangement. It uses a hexagonal prism unit cell and an ABABAB stacking sequence of close-packed hexagonal layers. Layer B sits in the triangular hollows of Layer A, and then Layer A repeats directly above the first Layer A.
Magnesium (Mg) | Titanium (Ti) | Zinc (Zn) | Cobalt (Co) | Zirconium (Zr) | Cadmium (Cd) | Beryllium (Be)
HCP metals generally have limited room-temperature ductility due to only 3 primary basal slip systems, compared to 12 in FCC — a key topic reviewed in ScienceDirect — HCP Materials Science.

6.2 Atoms per Unit Cell (HCP)
N = 12 corners × (1/6) + 2 face centres × (1/2) + 3 interior
= 2 + 1 + 3 = 6 atoms
6.3 Relationship Between a and R (HCP)
In the close-packed basal plane: a = 2R ⟹ R = a/2
Ideal c/a ratio = 1.633 (from close-packing geometry)
6.4 Volume of HCP Unit Cell
V_cell = (3√3/2) × a² × c
Using ideal c/a = 1.633 and a = 2R:
V_cell = (3√3/2) × (2R)² × (1.633 × 2R) = 33.94 R³
6.5 APF Derivation for HCP
Step 1: Volume of atoms
V_atoms = 6 × (4/3)πR³ = 8πR³ = 25.133 R³
Step 2: Volume of unit cell
V_cell = 33.94 R³
Step 3: Calculate APF
APF = 25.133 R³ / 33.94 R³ = 0.7405 ≈ 74.05%
7. Master Comparison — All Four Crystal Structures
Now that we have derived the packing fraction for all four major crystal structures, let us bring everything together. For deeper reading on each structure, see our tutorials on FCC, BCC, and HCP. Below is the APF visual comparison followed by the comprehensive data table — your definitive reference for examination preparation.
| Property | SC | BCC | FCC | HCP |
|---|---|---|---|---|
| Atoms / unit cell | 1 | 2 | 4 | 6 |
| Coordination number | 6 | 8 | 12 | 12 |
| Touching direction | Cube edge | Body diagonal | Face diagonal | In-plane edge |
| a vs R relationship | a = 2R | a = 4R/√3 | a = 2√2 R | a = 2R |
| APF (exact form) | π/6 | π√3/8 | π/(3√2) | π/(3√2) |
| APF (decimal) | 0.5236 | 0.6802 | 0.7405 | 0.7405 |
| APF (%) | 52.36% | 68.02% | 74.05% | 74.05% |
| Void fraction (%) | 47.64% | 31.98% | 25.95% | 25.95% |
| Close-packed? | No | No | Yes | Yes |
| Example metals | Polonium (Po) | Fe, Cr, W, Mo | Al, Cu, Au, Ni | Mg, Ti, Zn, Co |
| Typical ductility | Rare | Moderate | High | Low–Moderate |
SC (52%) < BCC (68%) < FCC = HCP (74%)
The more neighbours an atom has (higher coordination number), the higher the packing efficiency. Both FCC and HCP achieve the theoretical maximum — they are both called close-packed structures. The key difference between them is not packing efficiency but stacking sequence: ABCABC for FCC vs ABABAB for HCP.

8. Theoretical Density of Crystalline Materials
8.1 What is Theoretical Density?
The theoretical density of a crystalline material is the density calculated purely from its crystal structure data — the lattice parameter, the number of atoms per unit cell, and the atomic mass. It represents the density the material would have if the crystal were perfect — no vacancies, impurities, or grain boundaries.
In practice, the measured density of a real material is very close to (but slightly lower than) the theoretical density, because real crystals contain defects and voids. The theoretical density calculation is therefore a powerful check: if your calculated value matches the experimentally measured density, you can be confident your structural model is correct.
8.2 The Theoretical Density Formula
ρ = (n × A) / (V_c × N_A)
Where:
- n = atoms per unit cell (1 for SC, 2 for BCC, 4 for FCC, 6 for HCP)
- A = atomic mass (g/mol) — from the periodic table
- Vc = volume of the unit cell (cm³) — calculated from lattice parameter
- NA = Avogadro's number = 6.022 × 10²³ atoms/mol
The lattice parameter a is given in nm or Å in data tables. You MUST convert to cm before calculating Vc:
1 nm = 10⁻⁷ cm | 1 Å = 10⁻⁸ cm
Example: a = 0.2866 nm = 2.866 × 10⁻⁸ cm → Vc = (2.866 × 10⁻⁸)³ = 23.55 × 10⁻²⁴ cm³
9. Solved Problems — Step by Step
Step 1 — Count atoms in BCC unit cell:
N = 8 × (1/8) + 1 × 1 = 1 + 1 = 2 atoms
Step 2 — Atoms touch along body diagonal (length = a√3):
4R = a√3 ⟹ R = √3a/4
Step 3 — Calculate volumes:
V_atoms = 2 × (4/3)πR³ = (8/3)π × (√3a/4)³ = π√3 a³/8
V_cell = a³
Step 4 — APF:
APF = π√3 a³ / (8 a³) = π√3/8 = 3.14159 × 1.7321 / 8 = 0.6802 ✔
n = 2 (BCC)
a = 0.2866 nm = 2.866 × 10⁻⁸ cm
V_c = (2.866 × 10⁻⁸)³ = 23.55 × 10⁻²⁴ cm³
ρ = (2 × 55.85) / (23.55 × 10⁻²⁴ × 6.022 × 10²³)
ρ = 111.70 / 14.18 = 7.87 g/cm³ ✔
Experimentally measured density of iron = 7.87 g/cm³ — perfect agreement!
n = 4 (FCC)
a = 4.050 × 10⁻⁸ cm → V_c = (4.050 × 10⁻⁸)³ = 66.43 × 10⁻²⁴ cm³
ρ = (4 × 26.98) / (66.43 × 10⁻²⁴ × 6.022 × 10²³) = 107.92 / 40.00 = 2.698 g/cm³ ✔
Experimentally measured density of Al = 2.699 g/cm³ — essentially identical!
V_c = (n × A) / (ρ × N_A) = (4 × 63.55) / (8.94 × 6.022 × 10²³)
V_c = 254.2 / (5.384 × 10²⁴) = 4.722 × 10⁻²³ cm³
a = V_c^(1/3) = (47.22 × 10⁻²⁴)^(1/3) = 3.614 × 10⁻⁸ cm = 0.3614 nm
For FCC: 4R = a√2, therefore:
R = a√2 / 4 = 0.3614 × 1.4142 / 4 = 0.1278 nm = 127.8 pm ✔
Tabulated atomic radius of Cu = 128 pm — perfect match!
10. Practical Significance of Packing Fraction
10.1 Connection to Density
A higher APF means more atoms are packed into the same volume — the material is denser relative to its atomic mass. This is one reason why engineers choose between materials not just by chemistry, but by crystal structure.
10.2 Interstitial Voids and Diffusion
The void fraction (1 − APF) represents the empty interstitial spaces between atoms. These are critically important for several engineering phenomena:
- Carbon in steel: Carbon atoms occupy octahedral voids in iron's lattice. Carbon diffuses faster in BCC alpha-iron (void fraction 32%) than in FCC gamma-iron (void fraction 26%) — this is the basis of all steel heat treatment, as explained by DoITPoMS (University of Cambridge).
- Hydrogen embrittlement: Hydrogen atoms enter tetrahedral voids in BCC metals, causing catastrophic cracking in high-strength steels.
- Solid solution strengthening: Interstitial atoms in voids block dislocation motion — the fundamental mechanism of making iron into steel.
10.3 Allotropic Transformations and Density Changes
When iron transforms from BCC (alpha-iron, APF = 68%) to FCC (gamma-iron, APF = 74%) at 912°C, the unit cell volume decreases — atoms pack more efficiently. This volume change causes dimensional changes during heat treatment that engineers must account for in precision manufacturing.
11. Practice Multiple Choice Questions
- (a) Simple Cubic
- (b) BCC
- (c) FCC and HCP ✔ — both achieve 74.05%
- (d) Diamond cubic
- (a) Cube edge [100]
- (b) Body diagonal [111] ✔
- (c) Face diagonal [110]
- (d) They do not touch
- (a) 1
- (b) 2
- (c) 4 ✔ — FCC has 4 atoms per unit cell
- (d) 6
- (a) π/6
- (b) π√3/8
- (c) π/(3√2) ✔
- (d) π/4
- (a) a = 2R
- (b) a = 4R/√3 ✔
- (c) a = 2√2 R
- (d) a = R√3
- (a) 26%
- (b) 32% ✔ — since APF = 68%, void fraction = 100 − 68 = 32%
- (c) 48%
- (d) 15%
- (a) Iron
- (b) Copper
- (c) Polonium ✔
- (d) Magnesium
- (a) BCC iron has higher temperature
- (b) Carbon atoms are larger in BCC
- (c) BCC has a larger void fraction (32%) than FCC (26%) ✔
- (d) FCC iron has fewer unit cells
12. Key Takeaways — What You Have Learned
- APF DEFINITION: Fraction of unit cell volume occupied by atoms. APF = V_atoms / V_cell.
- SIMPLE CUBIC: 1 atom/cell, CN = 6, touches along cube edge (a = 2R), APF = π/6 = 52.36%.
- BCC: 2 atoms/cell, CN = 8, touches along body diagonal (4R = a√3), APF = π√3/8 = 68.02%.
- FCC: 4 atoms/cell, CN = 12, touches along face diagonal (4R = a√2), APF = π/(3√2) = 74.05%.
- HCP: 6 atoms/cell, CN = 12, touches in-plane (a = 2R), APF = 74.05% — same as FCC.
- HIERARCHY: SC (52%) < BCC (68%) < FCC = HCP (74%). Higher CN → higher APF.
- DENSITY FORMULA: ρ = (n × A) / (Vc × NA). Four variables: atoms/cell, atomic mass, unit cell volume, Avogadro's number.
- UNIT CONVERSION: Always convert a from nm to cm before calculating Vc. (1 nm = 10⁻⁷ cm)
- VOID FRACTION: (1 − APF) = empty space. Determines diffusion rate and interstitial solubility.
- VERIFICATION: Theoretical density must match experimental value — this confirms your structural model is correct.
-
W. D. Callister Jr. and D. G. Rethwisch, Materials Science and Engineering: An Introduction, 10th ed. Hoboken, NJ, USA: John Wiley & Sons, 2018, pp. 40–78.
[Publisher: Wiley] Primary textbook reference for APF derivations, unit cell geometry, theoretical density, and all four crystal structures (SC, BCC, FCC, HCP). -
J. F. Shackelford, Introduction to Materials Science for Engineers, 8th ed. Upper Saddle River, NJ, USA: Pearson Prentice Hall, 2015, pp. 52–95.
[Publisher: Pearson] Supporting textbook covering packing efficiency, coordination numbers, and interstitial void geometry in cubic and hexagonal structures. -
C. Kittel, Introduction to Solid State Physics, 8th ed. Hoboken, NJ, USA: John Wiley & Sons, 2005, ch. 1, pp. 1–26.
[Publisher: Wiley] Classic solid-state physics reference for Bravais lattice theory, hard sphere model, and APF calculations from first principles. -
International Union of Crystallography (IUCr), "Teaching Pamphlets: Crystal Symmetry and Space Groups," IUCr Education Pamphlets, no. 9. Chester, UK: IUCr, 2002.
[iucr.org — full text] Authoritative reference for the seven crystal systems and cubic crystal symmetry classifications used throughout this tutorial. -
A. Jain et al., "Commentary: The Materials Project: A materials genome approach to accelerating materials innovation," APL Mater., vol. 1, no. 1, Art. no. 011002, Jul. 2013, doi: 10.1063/1.4812323.
[BCC Fe crystal data — Materials Project mp-13] Source for verified lattice parameter and crystal structure data for iron (BCC), aluminium (FCC), and other metals used in solved problems. -
W. Pepperhoff and M. Acet, "The Structure of Iron," in Constitution and Magnetism of Iron and Its Alloys, Berlin, Germany: Springer, 2001, ch. 1, doi: 10.1007/978-3-662-04345-5_1.
[SpringerLink] Reference for iron allotropic transformations (BCC ↔ FCC) and the volume changes associated with the change in APF from 68% to 74% at 912°C. -
University of Cambridge, DoITPoMS, "Iron-Carbon Phase Diagram," Teaching and Learning Packages. Cambridge, UK: University of Cambridge, 2023.
[doitpoms.ac.uk] Peer-reviewed Cambridge academic resource on the physical basis of carbon solubility differences between BCC alpha-iron and FCC gamma-iron. -
ASM International, ASM Handbook, Volume 2: Properties and Selection — Nonferrous Alloys and Special-Purpose Materials. Materials Park, OH, USA: ASM International, 1990.
[asminternational.org] Industry-standard reference for the physical and mechanical properties of BCC refractory metals (W, Mo, Cr, Nb) and FCC metals (Al, Cu, Au, Ni). -
R. Verma and S. K. Rout, "Influence of annealing temperature on the existence of polar domain in uniaxially stretched polyvinylidene-co-hexafluoropropylene for energy harvesting applications," J. Appl. Phys., vol. 128, no. 23, Art. no. 234104, Dec. 2020, doi: 10.1063/5.0022463.
[AIP Publishing — DOI: 10.1063/5.0022463] Author's peer-reviewed research involving crystal structure analysis and phase identification in ferroelectric polymer systems. -
R. Verma and S. K. Rout, "Frequency-dependent ferro–antiferro phase transition and internal bias field influenced piezoelectric response of donor and acceptor doped bismuth sodium titanate ceramics," J. Appl. Phys., vol. 126, no. 9, Art. no. 094103, Sep. 2019, doi: 10.1063/1.5111505.
[AIP Publishing — DOI: 10.1063/1.5111505] Author's peer-reviewed research on crystal structure and phase analysis in perovskite materials, demonstrating the structural analysis methods discussed in this tutorial.
This tutorial is part of the Crystal Structure Master Hub at AdvanceMaterialsLab.com
Next in this series: Miller Indices | XRD & Bragg's Law | Defects in Crystals
© 2026 AdvanceMaterialsLab.com — All rights reserved
Dr. Rolly Verma
If you notice any inaccuracies or have constructive suggestions to improve the content, I warmly welcome your feedback. It helps maintain the quality and clarity of this educational resource. You can reach me at: advancematerialslab27@gmail.com
