Unit Cell and Lattice Parameters
The Fundamental Geometry of Crystals — Six Numbers That Describe Any Crystal in the Universe By Dr. Rolly Verma | AdvanceMaterialsLab.com | March 2026 | B.Sc. / M.Sc. Materials Science & EngineeringSeries: Crystal Structure Hub | Lecture: 03 of 10 | Prerequisites: Lecture 01 and Lecture 02 — Lattice and Basis
Reading time: 35 minutes | Includes: Unit cell concept, 6 lattice parameters, primitive vs conventional cells, Wigner-Seitz cell, XRD measurement, real crystal data table, worked examples, MCQs, key takeaways
SEO Keywords: unit cell and lattice parameters, unit cell crystallography, lattice constants, primitive cell vs unit cell, crystal lattice parameters a b c alpha beta gamma, Wigner-Seitz cell, XRD lattice parameter measurement
1. Connecting to Previous Lectures
Welcome back to the Crystal Structure Hub. So far in this series, we have built the two pillars of crystallographic description. In Lecture 01, we established that crystal structure — the periodic arrangement of atoms in space — governs the properties of materials, from the hardness of diamond to the conductivity of silicon. In Lecture 02, we introduced the mathematical language for describing this periodicity: the lattice and the basis, unified by the equation Crystal Structure = Lattice + Basis.
We know that the lattice is generated by three primitive vectors a₁, a₂, a₃. But a practical question now arises: when we are handed a real crystal — a piece of iron, a silicon wafer, a ferroelectric ceramic — how do we precisely characterise and communicate its geometric structure? We need a standardised, measurable framework. That framework is the unit cell and its six lattice parameters.
By the end of this lecture, you will be able to: (1) define the unit cell precisely and explain its power, (2) state and interpret all six lattice parameters, (3) distinguish between primitive and conventional unit cells and know when to use each, (4) identify which crystal system a material belongs to from its lattice parameters, and (5) understand how lattice parameters are measured experimentally and why they matter in engineering.
2. The Concept of the Unit Cell
2.1 The Tile Floor Analogy
Imagine walking into a large room with a beautifully patterned tile floor. The floor extends across thousands of square metres, yet it was designed by choosing just one tile and repeating it. Once you understand the design of that single tile — its shape, size, and pattern — you can reconstruct the entire floor. You do not need to study thousands of tiles; one is sufficient.
Crystals work in exactly the same way. Although a single crystal may contain 10²³ atoms or more, its entire structure is built by repeating a single, small structural unit over and over in three dimensions. That fundamental unit is the unit cell.
Think of building a large LEGO wall. You use identical bricks, placed edge to edge, in a regular repeating pattern. The entire wall — no matter how large — is fully described by the single LEGO brick's dimensions and the pattern of how it connects to its neighbours. The unit cell is that fundamental LEGO brick. The crystal is the wall.
The key insight: you do not need to describe every atom in a crystal separately. You need to describe only the unit cell — and the periodicity takes care of the rest.
2.2 Formal Definition
A unit cell is the smallest repeating parallelepiped (a three-dimensional box with six faces, each a parallelogram) that, when translated repeatedly along three independent directions using the lattice vectors, reproduces the entire crystal structure without gaps or overlaps.
Let us unpack each part of this definition carefully:
- "Smallest repeating" — the unit cell captures the minimum structural information needed. Larger cells are permissible (and often used for symmetry reasons, as we will see), but the defining requirement is repeatability.
- "Parallelepiped" — the unit cell is always a six-faced box. In the cubic crystal system it happens to be a perfect cube; in other systems it may be a skewed, non-rectangular box.
- "Translated repeatedly" — the crystal is generated by stacking copies of the unit cell side by side, with no empty spaces between them and no overlap. This is called space-filling tessellation.
- "Entire crystal structure" — the unit cell contains all the structural information: the positions of every atom type, the symmetry elements, and the geometry. Nothing is left out.
2.3 Why the Unit Cell Is So Powerful
The power of the unit cell concept is compression of information. Consider iron at room temperature. A single cubic centimetre of iron contains approximately 8.49 × 10²² atoms. Describing each atom separately would be impossible. But the entire structure is captured by one unit cell — a tiny cube of side length 0.2866 nm containing just 2 atoms, because iron adopts the BCC crystal structure.
From this single unit cell, materials scientists can calculate the density of iron, predict its X-ray diffraction pattern, determine its elastic constants, and model its electronic band structure. The unit cell is the atom of crystallography — the irreducible unit of structural information.
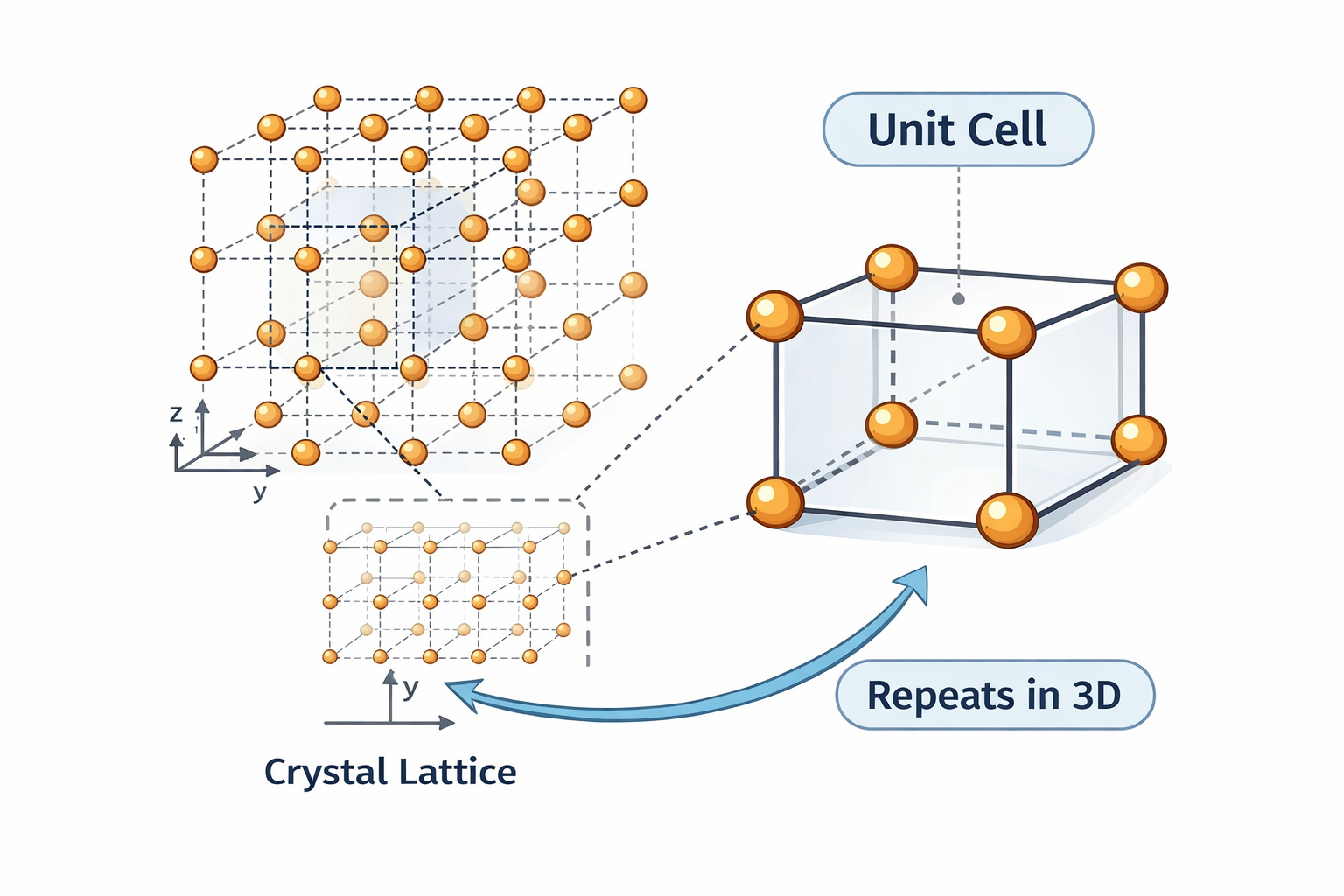
3. Six Lattice Parameters — The Complete Geometric Description
To fully specify the geometry of a unit cell, we need six numbers — the lattice parameters (also called lattice constants). These six numbers completely and unambiguously define the shape and size of any unit cell in any crystal system. They appear in every crystallographic database, every research paper, every structure determination report.
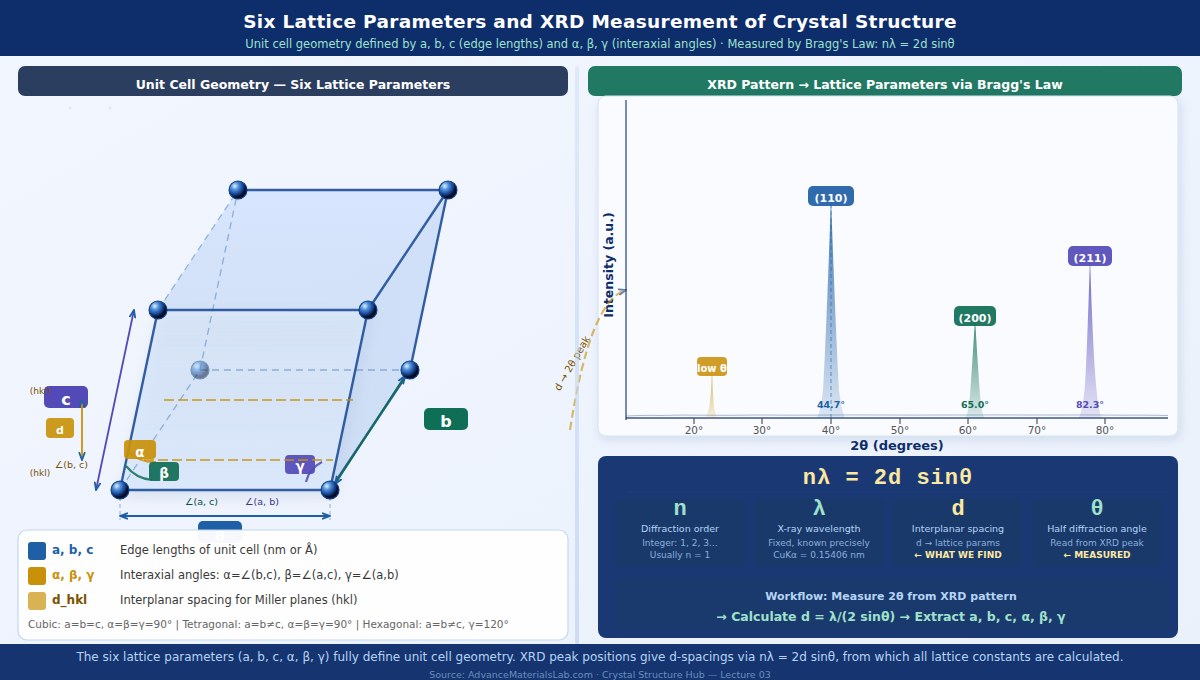
3.1 Edge Lengths: a, b, and c
The three edge lengths of the unit cell parallelepiped are denoted a, b, and c. They represent the physical distances between lattice points along the three independent directions in which the crystal repeats. Their units are typically nanometres (nm) or Ångströms (Å), where 1 Å = 0.1 nm = 10⁻¹⁰ m.
- a — the edge length along the x-direction (conventionally)
- b — the edge length along the y-direction
- c — the edge length along the z-direction
For high-symmetry systems, these may be equal (cubic: a = b = c) or partially equal (tetragonal: a = b ≠ c). For the lowest-symmetry triclinic system, all three are different.
3.2 Interaxial Angles: α, β, and γ
The three angles between the edge directions are denoted α, β, and γ:
α = angle between edges b and c (in the y-z plane)
β = angle between edges a and c (in the x-z plane)
γ = angle between edges a and b (in the x-y plane)
For the cubic, tetragonal, and orthorhombic systems, all angles are 90°. For the hexagonal system, γ = 120°. For the monoclinic system, one angle differs from 90°. For triclinic, all three are different and none needs to equal 90°.
3.3 Unit Cell Volume
The volume of a unit cell can be calculated from the six lattice parameters. For the general triclinic case, the full formula is:
V = abc √(1 − cos²α − cos²β − cos²γ + 2cosα·cosβ·cosγ)
This simplifies considerably for higher-symmetry systems. For the cubic system (a = b = c, α = β = γ = 90°):
V_cubic = a³
For the hexagonal system (a = b ≠ c, α = β = 90°, γ = 120°):
V_hexagonal = (√3/2) × a² × c = (3√3/2) × (a/√3)² × c
The unit cell volume directly determines the theoretical density of the material through the formula ρ = nA/(V × Nₐ), which is covered in full in our Packing Fraction and Theoretical Density tutorial.
4. The Seven Crystal Systems
The six lattice parameters can take many different relationships of equality and constraint. Mathematically, it turns out that all possible combinations reduce to exactly seven distinct crystal systems, as formally classified by the International Union of Crystallography (IUCr). Each system groups all crystal structures whose unit cells share the same symmetry constraints on their lattice parameters.
| Crystal System | Edge Lengths | Angles | Bravais Lattices | Example Material |
|---|---|---|---|---|
| Cubic | a = b = c | α = β = γ = 90° | SC, BCC, FCC | Fe, Cu, NaCl, Si |
| Tetragonal | a = b ≠ c | α = β = γ = 90° | Simple, Body-centred | In, TiO₂ (rutile), Sn |
| Orthorhombic | a ≠ b ≠ c | α = β = γ = 90° | 4 types | S, Ga, BaTiO₃ (RT phase) |
| Hexagonal | a = b ≠ c | α = β = 90°, γ = 120° | Simple | Mg, Zn, Graphite, ZnO |
| Rhombohedral | a = b = c | α = β = γ ≠ 90° | Simple | Bi, Calcite (CaCO₃) |
| Monoclinic | a ≠ b ≠ c | α = γ = 90°, β ≠ 90° | Simple, Base-centred | Gypsum, Feldspar, β-Se |
| Triclinic | a ≠ b ≠ c | α ≠ β ≠ γ ≠ 90° | Simple | Kaolinite, Turquoise, CuSO₄ |
The seven crystal systems are ordered by symmetry — from highest (cubic, with the most constraint) to lowest (triclinic, with no constraints). Cubic is the most symmetric because all three axes are equal and all angles are 90°. Triclinic is the most general because no two parameters need be equal. As you move down the hierarchy, you progressively relax constraints on the lattice parameters.
A powerful rule: any material must belong to exactly one crystal system. Measuring the six lattice parameters (by XRD) immediately tells you which system the material belongs to.
5. Primitive Cell vs Conventional Unit Cell
5.1 The Brick Wall Analogy
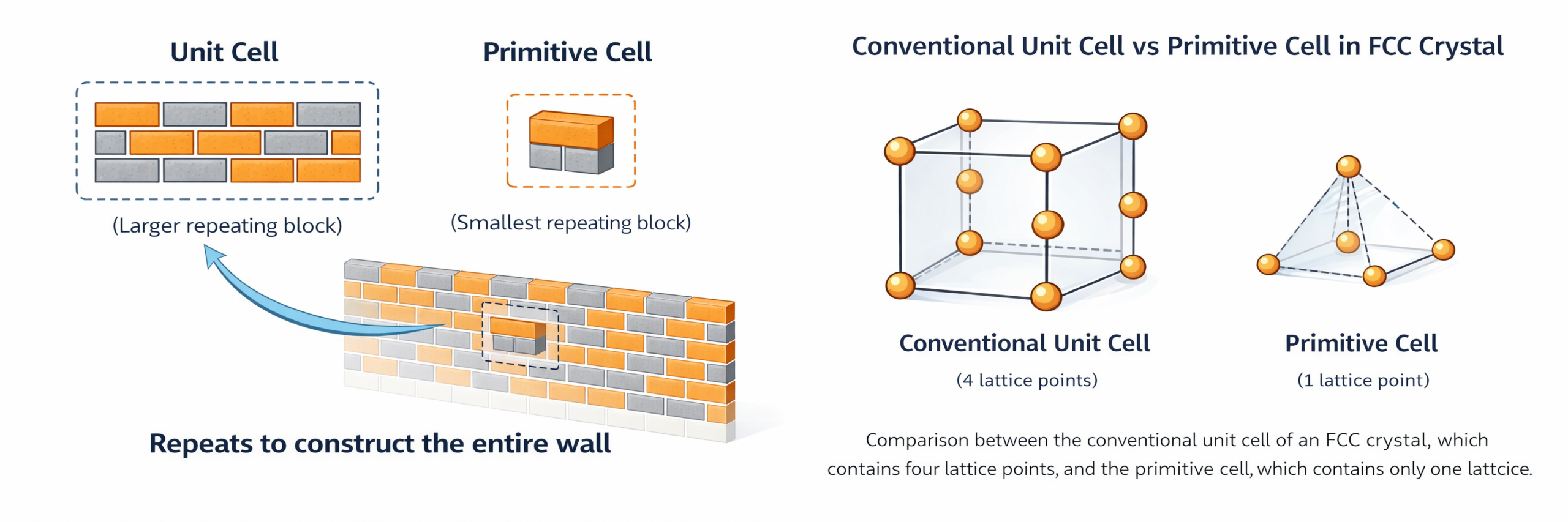
Consider a brick wall in a building. The wall is constructed from many bricks arranged in a repeating pattern — each row offset by half a brick width from the row below. To understand how the wall is built, you do not need to study every brick. You choose a representative block of the wall that captures the repeating pattern and can reproduce the whole wall when tiled. This block is your unit cell.
But now ask a more refined question: what is the smallest block that still captures the complete pattern? In the brick wall, you might find that a block containing just one brick, placed at an angle, is sufficient to tile the wall — even though the rectangular block you originally chose was easier to visualise. That smallest possible block is the primitive cell.
5.2 The Key Distinction
- Primitive cell: The smallest possible volume that can generate the entire crystal lattice by translation alone. Contains exactly one lattice point. Its shape may be an oblique parallelepiped — not always easy to visualise.
- Conventional unit cell: A unit cell chosen to clearly display the crystal's symmetry and is easy to work with. May contain more than one lattice point. Often larger than the primitive cell. In the cubic system, it is always a cube.
The crucial rule: every primitive cell is a unit cell, but not every unit cell is a primitive cell.
In practice, the two types of cell are used in different contexts:
- Conventional unit cells are used in introductory courses, experimental characterisation, and materials engineering because they clearly display the crystal symmetry and are easy to visualise. The BCC and FCC cells you encounter in textbooks are conventional cells.
- Primitive cells are preferred in advanced theoretical calculations — band structure computations, reciprocal lattice analysis, phonon dispersion — because they are more computationally efficient and contain the minimal repeating information.
5.3 The FCC Example — Four Lattice Points vs One
The face-centred cubic (FCC) structure is a perfect example of this distinction. The conventional FCC unit cell is a cube with atoms at the 8 corners and at the centre of each of the 6 faces. Counting using the sharing rule:
Conventional FCC unit cell: 8 corners × (1/8) + 6 face-centres × (1/2) = 1 + 3 = 4 lattice points
The conventional FCC cell contains 4 lattice points. It is four times larger than the primitive cell. However, the primitive cell of the FCC lattice contains exactly one lattice point. Its primitive vectors point from the cube corner to three nearest face-centre positions:
FCC primitive vectors:
a₁ = (a/2)(0, 1, 1)
a₂ = (a/2)(1, 0, 1)
a₃ = (a/2)(1, 1, 0)
FCC primitive cell volume = a³/4 = V_conventional / 4
The primitive cell of FCC is a rhombohedron — it contains one lattice point, but it does not look cubic. This is why we almost never use it in introductory courses — the conventional cubic cell is far more intuitive, even though it is four times larger.
6. Advanced Topic: The Wigner-Seitz Cell
There is a third type of unit cell that is extremely important in advanced solid-state physics and quantum materials research: the Wigner-Seitz cell. While it is not required knowledge for introductory courses, understanding it bridges the gap between crystallography and modern materials physics.
The Wigner-Seitz cell is a primitive cell constructed by a geometric procedure: it is the region of space that is closer to a given lattice point than to any other lattice point. It is constructed by drawing perpendicular bisector planes between a central lattice point and all its neighbours, then taking the enclosed volume.
The Wigner-Seitz cell has the same volume as the primitive cell and contains exactly one lattice point. But critically, it has the full point-group symmetry of the lattice — which the oblique primitive cell does not always display clearly. This symmetry makes the Wigner-Seitz cell the natural choice for physical calculations.
Its most important application is in reciprocal space: the Wigner-Seitz cell of the reciprocal lattice is called the first Brillouin zone — the fundamental domain for electronic band structure calculations. Every band structure diagram you see in semiconductor physics, with its k-space labels like Γ, X, L, K, is plotted within the first Brillouin zone, which is the Wigner-Seitz cell of the reciprocal lattice.
For the FCC direct lattice, the Wigner-Seitz cell is a truncated octahedron — a beautiful 14-faced polyhedron. For the BCC direct lattice, the Wigner-Seitz cell is a truncated octahedron as well (the reciprocal of FCC is BCC and vice versa).
7. Lattice Parameters of Real Crystals
Lattice parameters are not abstract numbers — they are precisely measurable physical quantities with direct engineering significance. The table below lists the experimentally measured lattice parameters of common engineering materials, verified against the Materials Project Database and standard crystallographic references.
| Material | Crystal System | Structure Type | a (nm) | c (nm) | Atoms / Cell |
|---|---|---|---|---|---|
| Iron (α-Fe) | Cubic | BCC | 0.2866 | — | 2 |
| Copper (Cu) | Cubic | FCC | 0.3615 | — | 4 |
| Aluminium (Al) | Cubic | FCC | 0.4050 | — | 4 |
| Silicon (Si) | Cubic | Diamond cubic | 0.5431 | — | 8 |
| Chromium (Cr) | Cubic | BCC | 0.2885 | — | 2 |
| Tungsten (W) | Cubic | BCC | 0.3165 | — | 2 |
| Magnesium (Mg) | Hexagonal | HCP | 0.3210 | 0.5210 | 2 |
| Titanium (Ti, α) | Hexagonal | HCP | 0.2951 | 0.4686 | 2 |
| Zinc (Zn) | Hexagonal | HCP | 0.2665 | 0.4947 | 2 |
| NaCl (table salt) | Cubic | Rock salt | 0.5640 | — | 8 |
| BaTiO₃ (tetragonal) | Tetragonal | Perovskite | 0.3994 | 0.4038 | 5 |
| Graphite (C) | Hexagonal | Layered AB | 0.2461 | 0.6708 | 4 |
Notice that BaTiO₃ has a tetragonal structure at room temperature with a ≠ c (a = 0.3994 nm, c = 0.4038 nm). The small but significant deviation c > a is not a measurement error — it is the physical signature of ferroelectric distortion. Above the Curie temperature (120°C), BaTiO₃ becomes cubic with a = c. The lattice parameter asymmetry directly measures the degree of ferroelectric distortion, as studied in Dr. Verma's own research on perovskite materials.
Also notice that silicon has a = 0.5431 nm but only 8 atoms per cell — a surprisingly large unit cell for a single-element material. This reflects the diamond cubic structure, where the large cell accommodates the tetrahedral bonding network.
8. How Lattice Parameters Are Measured — X-ray Diffraction
Lattice parameters are determined experimentally with extraordinary precision using X-ray diffraction (XRD). The physical principle is Bragg's Law:
nλ = 2d sinθ
Where:
n = diffraction order (integer: 1, 2, 3, ...)
λ = X-ray wavelength (fixed, known precisely — e.g. CuKα = 0.15406 nm)
d = interplanar spacing (distance between parallel crystal planes)
θ = half the diffraction angle (measured from the XRD pattern)
In a powder XRD experiment, the sample is illuminated with monochromatic X-rays and the intensity of scattered X-rays is recorded as a function of angle 2θ. Each peak in the pattern corresponds to diffraction from a specific set of crystal planes (hkl). The position of each peak gives the d-spacing for those planes through Bragg's Law, and from the d-spacings the lattice parameters are calculated using the appropriate formula for the crystal system.
For the cubic system, the d-spacing of the (hkl) plane family is:
1/d²_hkl = (h² + k² + l²) / a²
Therefore: a = λ√(h² + k² + l²) / (2 sinθ)
Modern XRD instruments can measure lattice parameters to a precision of ±0.0001 nm — four decimal places in nanometres. This extraordinary precision is what makes XRD the gold standard of crystal structure characterisation, as described in detail by the IUCr's guide to X-ray powder diffraction.
9. Worked Examples
In the BCC structure, atoms touch along the body diagonal. The body diagonal of the cube has length a√3. Along this diagonal, we have: corner atom (R) + body-centre atom (2R) + corner atom (R) = 4R. Therefore:
4R = a√3
R = a√3 / 4 = (0.2866 × 1.7321) / 4 = 0.4963 / 4 = 0.1241 nm = 124.1 pm
The experimentally measured atomic radius of iron is 124 pm — excellent agreement, confirming our BCC model is correct.
Check constraints systematically:
a = b ✓ (equal)
a ≠ c ✓ (0.399 ≠ 0.404)
α = β = γ = 90° ✓
This matches the tetragonal system (a = b ≠ c, all angles 90°). The material is barium titanate (BaTiO₃) in its room-temperature ferroelectric phase. The small but measurable difference between a and c (Δ = 0.005 nm, or 1.3%) is the structural signature of the ferroelectric distortion — the Ti⁴⁺ ion displaced off-centre along the c-axis.
FCC: n = 4 atoms per conventional unit cell
a = 0.3615 nm = 3.615 × 10⁻⁸ cm
V_cell = a³ = (3.615 × 10⁻⁸)³ = 47.27 × 10⁻²⁴ cm³
ρ = (n × A) / (V_cell × Nₐ)
= (4 × 63.55) / (47.27 × 10⁻²⁴ × 6.022 × 10²³)
= 254.2 / 28.47
= 8.93 g/cm³ ✔
Experimentally measured density of copper = 8.94 g/cm³ — essentially exact agreement.
10. Engineering Applications of Lattice Parameters
Lattice parameters are not merely academic quantities. They are measurable design variables in materials engineering with direct practical consequences across multiple industries.
10.1 Thin Film Epitaxy and Lattice Mismatch
When growing a crystalline thin film on a substrate (a process called epitaxy), the film's lattice parameters must closely match the substrate's. If they differ — a condition called lattice mismatch — the film is strained, and strain can dramatically modify the film's electronic, magnetic, or ferroelectric properties. The lattice mismatch parameter is defined as:
Lattice mismatch f = (a_film − a_substrate) / a_substrate × 100%
For the semiconductor industry, silicon-on-insulator wafers and III-V heterostructures (GaAs/AlAs, InGaAs/InP) are engineered with mismatches below 0.1% to achieve defect-free interfaces. The recently demonstrated topological crystalline insulator in bilayer SnTe on NbSe₂ (Nature Communications, 2025) achieved its quantum topological state specifically because compressive strain from the lattice-mismatched substrate modified the SnTe unit cell parameters, stabilising a topological phase.
10.2 Phase Transitions and Structural Monitoring
When materials undergo phase transitions — including the ferroelectric-to-paraelectric transition in BaTiO₃ at 120°C, the BCC-to-FCC transformation in iron at 912°C, and martensitic transformations in shape-memory alloys — the lattice parameters change discontinuously. Temperature-dependent XRD measurements track these parameter changes and precisely locate transition temperatures. This is a standard characterisation technique in functional materials research.
10.3 Residual Stress Measurement
When a metal component is machined, welded, or heat treated, internal stresses — called residual stresses — are introduced. These stresses slightly distort the crystal lattice, changing the lattice parameters by fractions of a percent. By measuring these distortions with high-resolution XRD, engineers can map the residual stress field in components non-destructively. This technique is used routinely in aerospace engineering, power generation, and automotive manufacturing to ensure component integrity, as documented in resources from ASM International.
10.4 Semiconductor Bandgap Engineering
In semiconductor device fabrication, the bandgap of a III-V semiconductor can be tuned by varying the alloy composition (e.g., In_xGa_{1-x}As), which changes the lattice parameter. This is the basis of bandgap engineering — the controlled adjustment of electronic properties by compositional and structural tuning. The relationship between composition, lattice parameter, and bandgap is described by Vegard's Law (linear interpolation of lattice parameters with alloy composition). Modern photonic devices, lasers, and photodetectors all exploit this principle.
11. Series Navigation
12. Practice Questions
- (a) It must always contain exactly one lattice point
- (b) It must be a perfect cube
- (c) It must tile all of space without gaps or overlaps when translated along lattice vectors ✔
- (d) It must contain at least two different atom types
- (a) Cubic ✔ — all edges equal, all angles 90°
- (b) Tetragonal
- (c) Hexagonal
- (d) Rhombohedral
- (a) 4 lattice points — same as the conventional cell
- (b) 1 lattice point ✔ — the primitive cell always contains exactly one lattice point
- (c) 2 lattice points
- (d) It depends on the material
- (a) The primitive cell
- (b) The conventional unit cell
- (c) The first Brillouin zone ✔ — fundamental domain for electronic band structure calculations
- (d) The Voronoi polyhedron
- (a) A measurement error in the XRD experiment
- (b) The material being amorphous rather than crystalline
- (c) Ferroelectric distortion — the off-centre displacement of Ti⁴⁺ breaks cubic symmetry ✔
- (d) The material being in the hexagonal crystal system
- (a) 0.1435 nm
- (b) 0.2866 nm ✔ — d(110) = λ/(2 sinθ) = 0.2027 nm, then a = d√(1²+1²+0²) = 0.2027×√2 = 0.2866 nm
- (c) 0.3615 nm
- (d) 0.4050 nm
13. Key Takeaways
- THE UNIT CELL: The smallest repeating parallelepiped that tiles all of space without gaps or overlaps when translated along lattice vectors. The fundamental building block of crystal structure — like a LEGO brick for the crystal world.
- SIX LATTICE PARAMETERS: Three edge lengths (a, b, c) and three interaxial angles (α, β, γ) completely and unambiguously describe the geometry of any unit cell in any crystal system.
- ANGLE DEFINITIONS: α is the angle between b and c; β between a and c; γ between a and b. In cubic systems, all angles are 90°.
- SEVEN CRYSTAL SYSTEMS: All possible unit cell geometries reduce to exactly 7 crystal systems — from the highest-symmetry cubic (a = b = c, α = β = γ = 90°) to the lowest-symmetry triclinic (no constraints).
- PRIMITIVE CELL: The smallest possible unit cell, containing exactly one lattice point. Always a unit cell, but has less intuitive shape (oblique parallelepiped).
- CONVENTIONAL UNIT CELL: Chosen to display crystal symmetry clearly; may contain more than one lattice point. Used in teaching, characterisation, and engineering.
- THE KEY RULE: Every primitive cell is a unit cell, but not every unit cell is primitive. FCC conventional cell has 4 lattice points; its primitive cell has 1.
- WIGNER-SEITZ CELL: A special primitive cell with full point-group symmetry. Its counterpart in reciprocal space — the first Brillouin zone — is the fundamental domain for electronic band structure calculations.
- XRD MEASUREMENT: Lattice parameters are measured experimentally with precision ±0.0001 nm using Bragg's Law (nλ = 2d sinθ). The peak positions in an XRD pattern directly encode the d-spacings and hence all six lattice parameters.
- ENGINEERING RELEVANCE: Lattice parameters govern residual stress, thin film epitaxy, phase transition temperatures, bandgap engineering, and ferroelectric distortion — making them primary design variables in modern materials engineering.
References
All references are in IEEE citation style. All sources are peer-reviewed journals, internationally recognised textbooks, or authoritative academic databases.
- C. Kittel, Introduction to Solid State Physics, 8th ed. Hoboken, NJ, USA: John Wiley & Sons, 2005, ch. 1, pp. 1–27. [Wiley] — Primary reference for unit cell geometry, primitive vectors, Wigner-Seitz cell construction, and Brillouin zone theory.
- N. W. Ashcroft and N. D. Mermin, Solid State Physics. Philadelphia, PA, USA: Holt, Rinehart and Winston, 1976, ch. 4–5. — Definitive graduate reference for primitive cells, Wigner-Seitz cells, reciprocal lattice, and the first Brillouin zone.
- W. D. Callister Jr. and D. G. Rethwisch, Materials Science and Engineering: An Introduction, 10th ed. Hoboken, NJ, USA: John Wiley & Sons, 2018, ch. 3. [Wiley] — Standard undergraduate reference for unit cell definition, lattice parameters, crystal systems, and XRD for lattice constant determination.
- B. D. Cullity and S. R. Stock, Elements of X-Ray Diffraction, 3rd ed. Upper Saddle River, NJ, USA: Pearson Prentice Hall, 2001. — Definitive reference for Bragg's Law, XRD lattice parameter measurement, d-spacing formulae for all crystal systems, and precision diffractometry.
- A. Kelly and K. M. Knowles, Crystallography and Crystal Defects, 2nd ed. Chichester, UK: John Wiley & Sons, 2012. [Wiley] — Reference for conventional vs primitive unit cells, lattice vector mathematics, and crystal system classification.
- International Union of Crystallography (IUCr), "Teaching Pamphlets: Crystal Symmetry and Space Groups," IUCr Education Pamphlets, no. 9. Chester, UK: IUCr, 2002. [iucr.org — full text] — Authoritative classification of the seven crystal systems, 14 Bravais lattices, and their lattice parameter constraints.
- IUCr, "Powder Diffraction," IUCr Teaching Pamphlets. Chester, UK: IUCr. [iucr.org — X-ray powder diffraction guide] — Authoritative reference for the experimental measurement of lattice parameters using X-ray powder diffraction.
- A. Jain et al., "Commentary: The Materials Project: A materials genome approach to accelerating materials innovation," APL Mater., vol. 1, no. 1, Art. no. 011002, 2013, doi: 10.1063/1.4812323. [Materials Project — open crystal database] — Source for all experimentally and computationally verified lattice parameters in Table 1 of this lecture.
- L. Jing et al., "Strain-induced two-dimensional topological crystalline insulator in bilayer SnTe," Nature Communications, vol. 16, Art. no. 6304, Jul. 2025, doi: 10.1038/s41467-025-67520-y. [Nature Communications — Open Access] — 2025 research demonstrating how strain-induced lattice parameter changes stabilise topological quantum phases — referenced in Section 10.1.
- R. Verma and S. K. Rout, "Frequency-dependent ferro–antiferro phase transition and internal bias field influenced piezoelectric response of donor and acceptor doped bismuth sodium titanate ceramics," J. Appl. Phys., vol. 126, no. 9, Art. no. 094103, Sep. 2019, doi: 10.1063/1.5111505. [AIP — DOI: 10.1063/1.5111505] — Author's research on perovskite crystal structure analysis, directly relevant to the BaTiO₃ lattice parameter discussion in Sections 7 and 10.2.
- R. Verma and S. K. Rout, "The Mystery of Dimensional Effects in Ferroelectricity," in Recent Advances in Multifunctional Perovskite Materials. London, UK: IntechOpen, 2022, doi: 10.5772/intechopen.104435. [IntechOpen — Open Access] — Author's chapter on how unit cell and lattice parameter changes drive ferroelectric phase transitions and size effects in perovskite materials.
This is Lecture 03 of the Crystal Structure Hub Series at AdvanceMaterialsLab.com
Next Lecture: BCC Crystal Structure — Complete Analysis →
© 2026 AdvanceMaterialsLab.com — Dr. Rolly Verma | All rights reserved
Dr. Rolly Verma
If you notice any inaccuracies or have constructive suggestions to improve the content, I warmly welcome your feedback. It helps maintain the quality and clarity of this educational resource. You can reach me at: advancematerialslab27@gmail.com
Explore More from Advance Materials Lab
Continue expanding your understanding of material behavior through our specialized resources on ferroelectrics and phase transitions. Each guide is designed for research scholars and advanced learners seeking clarity, precision, and depth in the field of materials science and nanotechnology.
🔗 Recommended Reads and Resources:
Ferroelectrics Tutorials and Research Guides — Comprehensive tutorials covering polarization, hysteresis, and ferroelectric device characterization.
Workshops on Ferroelectrics (2025–2027) — Upcoming training sessions and research-oriented workshops for hands-on learning.
Glossary — Ferroelectrics and Phase Transitions — Concise explanations of key terminologies to support your study and research work.
If you notice any inaccuracies or have constructive suggestions to improve the content, I warmly welcome your feedback. It helps maintain the quality and clarity of this educational resource. You can reach me at: advancematerialslab27@gmail.com
