Raman Spectroscopy: Principle, Working, Spectrum Interpretation and Applications in Ferroelectric and Piezoelectric Materials
A Complete Classroom Guide — From C.V. Raman's 200-Rupee Discovery to Phase Identification in BaTiO₃, PZT, BNT, PVDF and Graphene By Dr. Rolly Verma | AdvanceMaterialsLab.com | March 2026 | B.Sc. / M.Sc. / Ph.D. Materials Science, Physics & EngineeringSeries: Materials Characterisation Techniques | Prerequisite: Lecture 01 — XRD, Crystal Structure, Unit Cell & Lattice Parameters
Reading time: 55 minutes | Includes: C.V. Raman's story, what Raman spectroscopy is, Raman vs XRD vs FTIR, three scattering types, Raman shift and cm⁻¹ explained, Stokes vs Anti-Stokes, virtual state, spectrometer and laser selection, fluorescence, selection rules, mutual exclusion, how to read a spectrum, SERS, 5 ferroelectric case studies, GATE numericals, MCQs, IEEE references
SEO Keywords: what is Raman spectroscopy, Raman spectroscopy working principle, Raman vs FTIR vs XRD, Stokes anti-Stokes Raman scattering, what is Raman shift cm-1, virtual state Raman, PVDF beta phase Raman 839 cm-1, BaTiO3 Raman spectrum, BNT Raman modes, graphene D G 2D bands ID IG ratio, SERS surface enhanced Raman spectroscopy, Raman spectroscopy GATE questions
1. Introduction — C.V. Raman and a Discovery That Made India Proud
Let us begin this lecture with a story that every Indian scientist should know — and every scientist everywhere should admire.
It is February 1928. Chandrasekhara Venkata Raman, a 39-year-old physicist at the Indian Association for the Cultivation of Science in Calcutta, is conducting experiments on the scattering of light through liquids using nothing more than a mercury arc lamp and a primitive spectrograph. He notices that a tiny fraction of the scattered light has a slightly different colour — a different energy — from the incident light. The difference is not due to fluorescence (which he carefully rules out), but due to an entirely new physical phenomenon: the inelastic scattering of light by molecular vibrations.
On 28 February 1928, Raman and his student K.S. Krishnan publish the observation of what the world would soon call the Raman effect. Two years later, in 1930, C.V. Raman receives the Nobel Prize in Physics — the first Asian to win a Nobel Prize in science, and to this day the only person to have won it for an experiment conducted entirely in India, funded by Indian institutions, with Indian resources. The discovery was made with resources that cost less than 200 Indian rupees.
In honour of C.V. Raman's discovery on 28 February 1928, the Government of India declared 28 February as National Science Day. Every year on this day, India celebrates science and scientific achievement. The technique he discovered — Raman spectroscopy — has grown from a laboratory curiosity observed with sunlight and a mercury lamp into one of the most powerful, non-destructive analytical tools in modern science, used every day in laboratories from New Delhi to New York, from Munich to Melbourne.
As materials scientists, every time we record a Raman spectrum, we stand on the shoulders of this extraordinary Indian scientist.
In the previous lecture, we studied X-ray diffraction (XRD) — which tells us about long-range atomic order: which crystal phase is present, what the lattice parameters are, and whether the symmetry has changed. In this lecture, we study Raman spectroscopy — which tells us about local atomic bonding and vibration: how atoms are connected, how they move relative to each other, and whether the local chemical environment has changed. These two techniques are deeply complementary, and together they form the foundation of structural characterisation in materials science.
2. What is Raman Spectroscopy? The Answer in Plain Language
Before we go into any physics or equations, let me answer the single most searched question about this topic in one paragraph — in plain language that anyone can understand. For a concise technical overview from an instrument manufacturer's perspective, see also the Edinburgh Instruments guide to Raman spectroscopy.
Raman spectroscopy is a technique that identifies what a material is made of — and how its atoms are bonded and arranged — by shining a laser on it and analysing the light that comes back. When the laser light hits the material, the vast majority bounces back unchanged. But a tiny fraction of photons exchanges energy with the vibrating bonds inside the material and returns at a slightly different energy. The energy shift tells you which bonds are present and how they are vibrating — and since every material has a unique pattern of bond vibrations, this pattern is a molecular fingerprint. No sample preparation, no vacuum, no chemical damage. You point a laser at your sample and the bonds tell you who they are.
Now let me make this even more concrete. Suppose I give you a white powder and ask you to identify it. You cannot tell just by looking. You could dissolve it and run chemical tests — but that destroys the sample and takes hours. Alternatively, you could point a green laser at it for 30 seconds, collect the Raman spectrum, compare it to a database, and know exactly what it is — all without touching it, dissolving it, or changing it in any way. That is Raman spectroscopy in everyday practice.
What can it tell you specifically? Here is the list that matters for our work:
- Chemical identity: Which compound is this? Match the spectrum to a database and identify the material — pharmaceutical, mineral, polymer, ceramic, or biological tissue.
- Crystal phase: Which crystallographic phase is present? Tetragonal or cubic BaTiO₃? α-phase or β-phase PVDF? Raman identifies phase transitions with extraordinary sensitivity.
- Bond type: What types of bonds exist? C-C, C=C, Ti-O, Bi-O, O-H? Each bond vibrates at a characteristic frequency and appears at a characteristic cm⁻¹ position.
- Crystallinity and disorder: How well-ordered is the material? Sharp peaks mean high crystallinity; broad peaks mean disorder, nanocrystallinity, or amorphous character.
- Stress and strain: Is the material under mechanical stress? Compressive or tensile strain shifts Raman peaks predictably — and Raman is used to map stress distributions in semiconductor devices and thin films.
- Temperature: What is the local temperature of a small spot? The ratio of Anti-Stokes to Stokes intensity gives temperature without contact — used inside running transistors and inside living cells.
- Works on practically anything: Solids, powders, liquids, gels, films, gases, biological tissue, samples through glass containers — Raman is remarkably versatile.
There is one important limitation to mention upfront: Raman gives you local information — the bonding environment within a few nanometres of the laser focus. It does not give you the long-range periodic crystal structure that XRD provides. This is not a weakness — it is a different and complementary kind of information. And for detecting subtle phase transitions in ferroelectric materials, where the symmetry change is local before it is global, Raman often sees the transition before XRD does.
3. Raman vs XRD vs FTIR — When to Use Which Technique
The second most searched question about Raman spectroscopy is: "How is it different from FTIR and XRD?" This is exactly the right question to ask — and the answer is what determines which instrument you reach for when you walk into the characterisation lab. A detailed technical comparison is also available at the Renishaw guide to Raman spectroscopy.
Let me use a simple analogy first. Imagine you have a new synthetic material and you want to characterise it completely. Think of XRD, Raman, and FTIR as three different doctors examining the same patient:
- XRD is the bone X-ray — it shows the overall skeletal structure, the long-range periodic arrangement of atoms, the distances between planes. Excellent for identifying crystal phases and measuring lattice parameters. Cannot detect amorphous regions and needs crystallinity.
- Raman is the MRI scan — it probes the local chemical environment, the bonds, the local vibrations, whether a particular structural unit is intact. Works on both crystalline and amorphous materials. Excellent for detecting subtle symmetry changes that an X-ray might miss.
- FTIR is the blood test — it detects specific functional groups and molecular species through infrared absorption. Excellent for organic materials and functional group identification. Hampered by water interference.
| Property | XRD | Raman Spectroscopy | FTIR Spectroscopy |
|---|---|---|---|
| What it probes | Long-range crystal structure | Local bond vibrations (phonons) | Bond vibrations via IR absorption |
| Needs crystallinity? | Yes — amorphous gives only broad halo | No — works on crystalline AND amorphous | No — works on both |
| Water sensitivity | Not affected | Excellent — water is a very weak Raman scatterer | Poor — water strongly absorbs IR |
| Sample preparation | Moderate — flat, homogeneous surface needed | Minimal — analyse as received, through glass | Moderate — KBr pellet or ATR contact |
| Spatial resolution | Bulk average (mm scale) | ~1 μm with confocal microscope | ~10 μm with IR microscope |
| Amorphous detection | Weak — broad hump only | Good — broad Raman features appear | Good — IR bands still present |
| Phase transitions | Peak splitting, position shift | New modes appear, mode softening | Band shifts, limited symmetry info |
| Selection rule | Systematic absences (lattice type) | Polarisability change (∂α/∂Q ≠ 0) | Dipole moment change (∂μ/∂Q ≠ 0) |
| Best for our research | Phase ID, lattice parameters, crystallite size | Phase transitions, local bonding, BNT/PVDF/PZT identification | Polymer phase (PVDF α/β), organic ligands |
For ferroelectric and piezoelectric research, the most powerful approach is to use all three techniques on the same sample. XRD confirms long-range crystal structure and phase purity. Raman reveals local bonding changes and phase transitions that XRD may miss. FTIR confirms polymer phases (especially PVDF α vs β) and organic functional groups. Each technique covers what the others cannot see. The best published papers in ferroelectric materials characterisation always use at least two of these three techniques — and most leading groups use all three.
4. What Happens When Light Hits a Molecule — Three Possible Outcomes
Imagine you are throwing a tennis ball at a spinning fan. Most of the time, the ball bounces straight back at the same speed — same energy, no change. Occasionally, the ball catches a blade at just the right moment and bounces back either faster or slower than it went in, depending on whether it gained or lost energy from the spinning blade.
That is exactly what happens when a laser photon hits a vibrating molecule. The photon arrives, interacts with the molecule's electron cloud for a fraction of a nanosecond, and one of three things happens — simultaneously, just at very different probabilities. A clear visual explanation of this process is available at Renishaw's basic overview of Raman scattering.
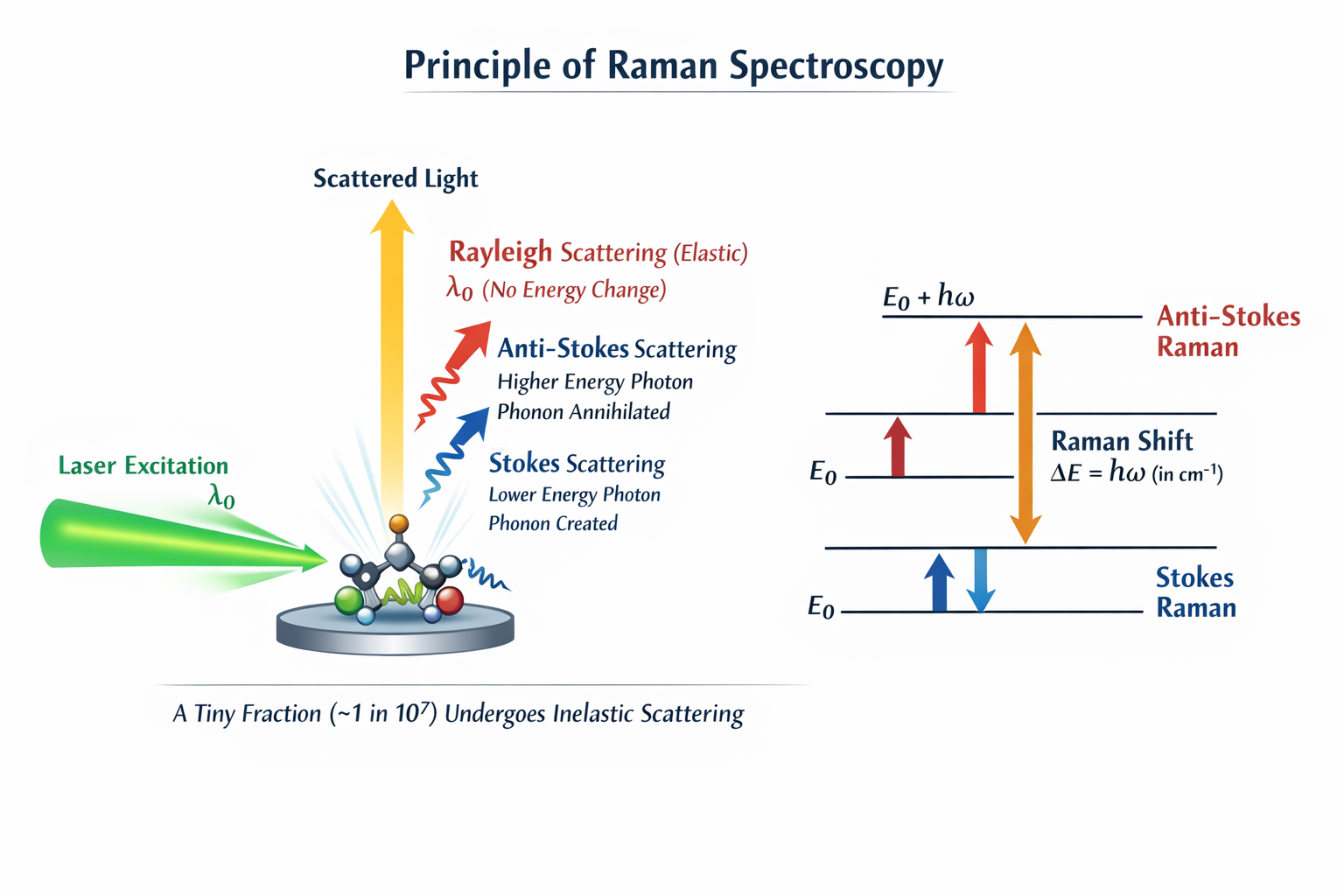
① Rayleigh Scattering — The "Safe Bounce" (999,999 out of every 1,000,000 photons)
The photon hits the molecule, temporarily disturbs its electron cloud, and bounces back with exactly the same energy it arrived with. Nothing is exchanged. The molecule is left in exactly the same vibrational state as before. This is elastic scattering — like a perfectly elastic collision in physics. Because it carries no chemical information (the photon left with the same energy it brought), it is useless for analysis. But it is so intense — millions of times stronger than the Raman signal — that a special filter called a notch filter or edge filter is placed in every Raman spectrometer specifically to block it before the detector. Without this filter, the Rayleigh signal would blind the detector completely.
② Stokes Raman Scattering — The Photon "Donates" Energy (about 1 in 1,000,000 photons)
Here is where it gets chemically interesting. Occasionally, the photon arrives at a molecule sitting quietly in its lowest energy state (the ground state). As the photon passes through, it excites a vibrational mode — it kicks the molecule into oscillation, like plucking a guitar string. The photon pays for this by donating some of its own energy. It then leaves with less energy than it arrived with — a lower frequency, a longer wavelength, a redder colour. The energy difference between the incoming and outgoing photon is exactly the vibrational energy of the bond that was excited. This is a Stokes Raman photon. It appears on the right side of the laser line in the spectrum. This is what every Raman experiment measures.
③ Anti-Stokes Raman Scattering — The Photon "Steals" Energy (even rarer)
Now imagine a molecule that is already vibrating — it already has some extra thermal energy because room temperature provides enough heat to excite a small fraction of bonds into their first excited vibrational state. When a photon hits this already-vibrating molecule, it can absorb that vibration — literally stealing the molecule's thermal energy — and leave with more energy than it brought. Higher energy means higher frequency, shorter wavelength, bluer colour. This is an Anti-Stokes Raman photon. It appears on the left side of the laser line. Anti-Stokes lines are always weaker than Stokes because at room temperature, most molecules are in the ground state — only a small fraction are already vibrating. The hotter your sample, the stronger the Anti-Stokes signal. This fact is exploited in Raman thermometry — measuring temperature without contact.
A critical point for your lab work: when you record a Raman spectrum, the x-axis starts at zero (the laser line position, which is blocked by the filter) and increases to the right. Everything you see is Stokes peaks. The cm⁻¹ values you read off the x-axis are the energy the photons gave to the material — the fingerprints of its bonds.
5. The Raman Shift and cm⁻¹ — The X-axis Demystified Step by Step
Let me take a moment here, because this is the concept that trips up almost every student when they look at a Raman spectrum for the first time. They see the x-axis labelled "cm⁻¹" and immediately ask — what is that? Is it a wavelength? Is it a frequency? Why not just use nanometres like everywhere else?
Let us answer this carefully and completely.
Step 1 — What is a Wavenumber?
Forget Raman for a moment. A wavenumber (symbol ν̃, pronounced "nu-tilde") simply tells you how many complete wave cycles fit into one centimetre of space. It is an alternative way to express the frequency of light, but in a unit that spectroscopists prefer because it is directly proportional to energy:
Wavenumber ν̃ = 1/λ (in cm⁻¹, where λ is the wavelength in cm)
Examples:
532 nm = 532 × 10⁻⁷ cm → ν̃ = 1/(532 × 10⁻⁷) = 18,797 cm⁻¹
785 nm = 785 × 10⁻⁷ cm → ν̃ = 1/(785 × 10⁻⁷) = 12,739 cm⁻¹
Higher wavenumber = higher energy = shorter wavelength
Higher wavenumber means higher energy — the opposite of wavelength. Once you accept this, cm⁻¹ becomes very natural. Chemists and spectroscopists prefer it precisely because it is proportional to energy, making it easy to compare bond vibration energies directly.
Step 2 — Why Not Just Plot the Absolute Wavelength of the Scattered Light?
Here is the problem. When a Raman photon is scattered, it arrives at the detector at some absolute wavelength — say, 548 nm if you used a 532 nm laser, or 803 nm if you used a 785 nm laser. But here is the thing: the same Ti-O bond in BaTiO₃ scattering at 515 cm⁻¹ would produce scattered photons at completely different absolute wavelengths depending on which laser you used. Two labs, two lasers, two different absolute wavelength positions — yet they are measuring exactly the same bond.
If we plotted absolute wavelength on the x-axis, the same material would produce peaks at different positions in different labs. Comparison would be impossible. So instead, we plot the Raman shift Δν̃ — the energy the photon lost to the material, expressed in cm⁻¹:
Raman Shift Δν̃ = ν̃_laser − ν̃_scattered
Converting from wavelengths in nanometres:
Δν̃ = ( 1/λ_laser − 1/λ_scattered ) × 10⁷ [λ in nm → Δν̃ in cm⁻¹]
Worked example — Ti-O mode of BaTiO₃ with 532 nm laser:
If the scattered photon arrives at 541.5 nm:
ν̃_laser = 18,797 cm⁻¹
ν̃_scattered = 18,466 cm⁻¹
Δν̃ = 18,797 − 18,466 = 331 cm⁻¹ ← this is the x-axis value you read
The Raman shift Δν̃ is completely independent of the excitation laser wavelength. Whether your lab in Ranchi uses a 532 nm laser, a collaborator's lab in Tokyo uses a 785 nm laser, or a synchrotron facility uses 650 nm — the Ti-O stretching mode of BaTiO₃ will always appear at 515 cm⁻¹ in all three labs. Always. The absolute wavelengths of the scattered photons will be completely different, but once converted to Raman shift, they are identical.
Why? Because the Raman shift equals the actual vibrational energy of the bond being probed. A Ti-O bond vibrates at a fixed frequency determined by the masses of Ti and O and the strength of their bond — not by the colour of the laser. The laser is just the probe. What you are measuring is the material's own vibrations.
This is why Raman spectroscopy is called a molecular fingerprint technique — the pattern of peaks in cm⁻¹ is as unique to a material as your fingerprint is to you. Two different pieces of BaTiO₃ from two different continents, measured with two different lasers in two different labs, will produce the same Raman spectrum. That is both beautiful and scientifically fundamental.
To summarise in one sentence: the x-axis of a Raman spectrum shows how much energy each scattered photon lost to the material's vibrations — expressed in the convenient unit of cm⁻¹ — and this number tells you everything about the material and nothing about the laser.
6. Why We Always Measure Stokes Lines — The Boltzmann Story
You now know that both Stokes and Anti-Stokes Raman peaks exist on either side of the laser line. So here is the natural question: if both exist, why does every Raman spectrum you have ever seen only show the Stokes side? Why not use Anti-Stokes?
The answer comes from a concept you may remember from thermodynamics — the Boltzmann distribution. Let me connect it to what we just learned.
Think of a molecule as a tiny vibrating spring. At absolute zero (0 K), the spring is completely still — the molecule is in its ground vibrational state (v = 0). At room temperature, thermal energy kicks a fraction of molecules up into their first excited vibrational state (v = 1). Not all of them — just a fraction determined by how large the vibrational energy is compared to the thermal energy available (kT).
The fraction of molecules in the excited state compared to the ground state is:
N₁/N₀ = exp(−hcΔν̃/kT)
h = Planck's constant = 6.626 × 10⁻³⁴ J·s
c = speed of light = 2.998 × 10¹⁰ cm/s
Δν̃ = Raman shift of the mode (cm⁻¹)
k = Boltzmann constant = 1.381 × 10⁻²³ J/K
T = temperature in Kelvin (room temperature = 298 K)
Now the key connection: Stokes scattering needs molecules in v = 0 (very abundant at room temperature). Anti-Stokes scattering needs molecules in v = 1 (much rarer). The intensity ratio is:
I_Anti-Stokes / I_Stokes ≈ exp(−hcΔν̃/kT)
For the Ti-O mode of BaTiO₃ at Δν̃ = 500 cm⁻¹, T = 298 K:
exp(−hc×500/kT) = exp(−2.41) ≈ 0.09
Anti-Stokes intensity is only 9% of Stokes intensity at room temperature.
For a mode at 800 cm⁻¹: it drops to ~2%.
For a C-C stretch at 1500 cm⁻¹: it drops to less than 0.1%.
Why we always use Stokes: Stokes peaks are always much stronger — anywhere from 10× to 1000× stronger depending on the vibrational frequency. Using Stokes gives clearer spectra with better signal-to-noise. By international convention, all published Raman spectra plot Stokes shifts (positive cm⁻¹ values on the x-axis).
The hidden superpower of Anti-Stokes — Raman thermometry: Look at the equation again — the ratio I_AS/I_S depends on temperature T. If you measure both sides of the Raman spectrum in a running experiment and calculate the ratio, you can directly compute the local temperature of that exact spot — without any thermocouple, without contact, in real time. This is called Raman thermometry and it is used to measure temperature inside running transistors (where no thermocouple fits), inside biological cells (where contact would kill the cell), and inside materials under extreme pressure in diamond anvil cells. The hotter the sample, the stronger the Anti-Stokes signal becomes — at 1000°C, Anti-Stokes peaks are easily visible and measurable. That is not a textbook curiosity. It is active research.
7. What is a Virtual State? — The Most Searched Raman Question
Every student who studies for GATE, CSIR-NET, or IIT JAM Physics encounters this question: "What is the virtual state in Raman spectroscopy?" And almost every textbook answers it with a Jablonski diagram and a few sentences that leave the student more confused than before.
Let me explain it from scratch — no diagrams needed, just a clear mental picture.
First, Let Us Clarify What We Mean by "Energy States"
You know that molecules have discrete energy levels — the ground state (lowest energy), and excited states at higher energies. In fluorescence, a molecule absorbs a photon that matches exactly the energy gap between the ground state and an excited state. The molecule actually goes to that excited state, dwells there for nanoseconds, and then emits a photon as it returns.
The key phrase is "matches exactly." Fluorescence requires the photon energy to match a real, stable energy level of the molecule.
What is Different About Raman?
In Raman scattering, the laser photon typically does NOT match any real electronic energy level of the molecule. So where does it go? This is the question that the virtual state answers.
Imagine the photon arrives and briefly distorts the electron cloud of the molecule — squeezing and stretching it momentarily. During this distortion, the molecule is in an extremely unstable, transient configuration that does not correspond to any real energy level. This is the virtual state. It exists only for an unimaginably short time — on the order of 10⁻¹⁴ seconds (10 femtoseconds). The molecule cannot stay there. It must immediately emit a photon and return to a stable state.
Imagine you are playing catch. A ball (the photon) comes toward you. In normal absorption (fluorescence), you catch the ball and hold it — the ball has entered a real, stable state in your hands. You hold it for a while (nanoseconds) and then throw it back.
In Raman scattering, the ball arrives but you cannot properly grip it — your hands are the wrong shape for this particular ball. It touches your hands for a fraction of a second (femtoseconds), barely makes contact, and immediately bounces away. During that brief contact, a tiny amount of energy was exchanged between you and the ball — you either gave a little spin to the ball (Stokes: ball loses energy) or absorbed some spin from it (Anti-Stokes: ball gains energy). But you never truly held it. The virtual state is the momentary, unstable contact — not a proper grip.
This is why Raman scattering can happen with almost any laser wavelength, while fluorescence only happens when the laser matches a specific molecular absorption. Raman does not need a real energy match. The virtual state is not a real energy level — it is just a description of the fleeting, transient distortion of the electron cloud as the photon passes through.
Why Does the Virtual State Matter for Raman?
Because the virtual state is unstable and the molecule immediately emits a photon, two outcomes are possible:
- The molecule returns to the same vibrational state it started from → the emitted photon has the same energy as the incoming photon → Rayleigh scattering
- The molecule returns to a different vibrational state (higher or lower) → the emitted photon has a different energy → Raman scattering (Stokes or Anti-Stokes)
The probability of the second outcome — Raman scattering — is extremely small (1 in 10⁷) precisely because the virtual state is so unstable. The molecule almost always just bounces the photon back unchanged (Rayleigh).
What Happens When the Laser Matches a Real State? — Resonance Raman
Here is a natural extension of this concept that GATE and CSIR-NET frequently test. Suppose you choose a laser wavelength whose photon energy exactly matches a real electronic transition of the molecule. Now instead of passing through an unstable virtual state for 10 femtoseconds, the electron actually enters a real, stable excited state where it can linger.
When the laser energy matches a real electronic transition, the probability of Raman scattering increases enormously — by factors of 10² to 10⁶. This is called Resonance Raman Spectroscopy (RRS). You are using the same instrument, the same setup — just a laser wavelength tuned to the material's electronic absorption.
Think of it like the swing analogy: normally you push the swing at random times and it barely moves (non-resonant Raman). Tune your push frequency to the swing's natural frequency and the amplitude builds dramatically (resonance). The physics is the same — resonance always amplifies.
Where is this used? In characterising haemoglobin (the red colour of blood comes from an electronic absorption at ~540 nm — a green laser resonates with it and dramatically enhances specific vibrational modes), conjugated polymers (π→π* transitions in the visible range), chlorophyll in photosynthesis research, and most famously in single-walled carbon nanotubes — where each tube diameter has its own characteristic electronic transition. Choose the right laser and you selectively light up one tube species from a mixture of thousands, making it visible while others remain nearly silent.
8. The Raman Spectrometer — How the Machine Works
Now that you understand what Raman scattering is and why it happens, let us look at the instrument that detects it. A modern Raman spectrometer is conceptually simple — it has four essential components arranged in a specific sequence, each one solving a specific problem. For a well-illustrated guide to Raman instrumentation, see the Bruker guide to Raman spectroscopy basics.
Let me walk you through it as if we were building one from scratch:
Problem 1: We need a pure, single-frequency light source. Solution: a laser. Lasers produce highly monochromatic, coherent, high-intensity light at a precisely known wavelength. The intensity is critical because Raman scattering is so weak — we need as many photons as possible hitting the sample per second.
Problem 2: The Rayleigh scattered light is millions of times stronger than the Raman signal and will overwhelm the detector. Solution: a notch filter or edge filter placed immediately after the sample. This filter blocks a narrow band of wavelengths around the laser line with extremely high efficiency, while passing all the slightly shifted Raman photons. Without this filter, detecting Raman would be like trying to see a candle flame next to the sun.
Problem 3: The Raman photons arrive at many different wavelengths (one for each vibrational mode) and we need to separate them. Solution: a diffraction grating inside a spectrograph. The grating diffracts different wavelengths at different angles, spreading them spatially — shorter wavelengths bend more, longer wavelengths bend less. This converts a spectrum of wavelengths into a spatial distribution across the detector.
Problem 4: We need to record the intensity at each wavelength simultaneously and with very low noise. Solution: a CCD detector (Charge-Coupled Device — the same type of sensor used in high-quality cameras). Modern CCD chips can detect individual photons. They record the entire spectrum simultaneously, making measurements fast and sensitive.
The standard laboratory configuration is 180° backscattering geometry: the laser is focused onto the sample through a microscope objective, and the scattered light is collected by the same objective and directed backward through the filter and into the spectrograph. This confocal geometry — with a small pinhole that rejects light from above and below the focal plane — allows measurements from volumes as small as 1 cubic micrometre. This is micro-Raman spectroscopy — the ability to record a Raman spectrum from a spot smaller than a red blood cell.
8.1 Choosing the Right Laser: 532 nm, 785 nm, or 1064 nm?
This is a decision every Raman spectroscopist makes before every experiment. The laser wavelength affects the signal strength, fluorescence background, sample heating, and detector efficiency. Three wavelengths dominate laboratory use. For a comprehensive overview of Raman instrumentation and laser considerations, the HORIBA Raman spectroscopy resource centre is an excellent reference.
| Laser | Excitation Efficiency | Fluorescence Risk | Best Suited For |
|---|---|---|---|
| 532 nm (green) | Highest — λ⁻⁴ law means 4.7× more efficient than 785 nm | Highest — can excite most organic fluorophores | Inorganic ceramics (BaTiO₃, BNT, PZT), metal oxides, carbon allotropes. Full spectral range 65–4000 cm⁻¹. |
| 785 nm (NIR) | Medium — 4.7× less than 532 nm | Low — avoids most fluorophores | Most versatile choice. Works for 90% of materials. Best for polymers, pharmaceuticals, biological samples, ceramics with organic contamination. |
| 1064 nm (NIR) | Lowest — 16× less than 532 nm | Lowest — fluorescence essentially eliminated | Strongly fluorescent samples: dyes, oils, coloured polymers, plant materials. Last resort when 785 nm still shows fluorescence. |
The efficiency difference comes from the physics of Raman scattering — the signal intensity is proportional to the fourth power of the excitation frequency (I ∝ ν⁴ ∝ λ⁻⁴). This means halving the wavelength (going from 1064 nm to 532 nm) increases the Raman signal by a factor of 2⁴ = 16 times. That is a significant gain — but it comes at the cost of higher fluorescence risk and sometimes sample photodegradation.
For our ferroelectric research: start with 532 nm for ceramic powders (BaTiO₃, BNT, PZT) — they rarely fluoresce, and the higher efficiency gives cleaner spectra faster. Switch to 785 nm for PVDF and other polymers, which often fluoresce at shorter wavelengths due to residual solvents or processing agents.
8.2 Fluorescence — The Most Frustrating Problem in Raman Spectroscopy
If you have ever collected a Raman spectrum and seen a broad, sloping, featureless background that rises dramatically and buries all your peaks — you have experienced fluorescence interference. It is the most common practical problem in Raman spectroscopy, and every student needs to know how to diagnose it and fix it.
Fluorescence happens when the laser photon is absorbed by an electronic transition in the sample (or, more commonly, in a tiny fluorescent impurity) and the excited electron relaxes by emitting a broad band of photons across a wide wavelength range. This emission can be 10⁶ to 10⁹ times more intense than the Raman signal — and it completely buries the weak Raman peaks beneath its enormous background.
The diagnostic test: Change the laser wavelength. Raman peaks will stay at exactly the same cm⁻¹ positions (because they measure the material's bond energies, not the laser). Fluorescence will shift in cm⁻¹ position (because it is anchored at a fixed absolute wavelength, so changing the laser changes its apparent cm⁻¹ position).
Solutions (try in this order):
- Switch to 785 nm laser — this alone solves fluorescence in 70% of cases. NIR light excites fewer electronic transitions and avoids most fluorophores.
- Photo-bleach the sample — leave the laser focused on the sample for several minutes before recording the spectrum. Fluorescent molecules absorb the laser and are photo-chemically destroyed. The fluorescence background decays while Raman peaks remain stable.
- Clean the sample surface — fluorescence often comes from surface contamination (residual solvents, organic films, fingerprints) rather than the bulk material. Cleaning with ethanol can eliminate it.
- Switch to 1064 nm laser — virtually eliminates fluorescence but at the cost of 16× weaker signal and longer measurement times.
- Polynomial baseline correction in software — fit a polynomial curve to the background and subtract it. This is a mathematical correction rather than a physical solution, but it works when the fluorescence is smooth and gradual.
9. Selection Rules — Why Some Vibrations Are Raman Active and Others Are Not
Here is a question that might have already occurred to you. A crystal like BaTiO₃ has 5 atoms per formula unit, and a large unit cell has hundreds of atoms vibrating in all kinds of patterns. Does every single vibration show up as a peak in the Raman spectrum? If so, the spectrum would be an impenetrable forest of peaks.
The answer is no — and the elegant mathematical framework of group theory tells us exactly which vibrations appear in the Raman spectrum, before we ever run a single experiment. The selection rule is based on a simple physical principle that you can understand without any group theory at all.
The Core Idea: Polarisability Must Change
Think of a molecule's electron cloud as a balloon. Some vibrations cause this balloon to expand and contract symmetrically as the molecule oscillates — like a balloon breathing. This deformation of the electron cloud shape is called a change in polarisability (symbol α). If the balloon changes shape during a vibration, the vibration is Raman active — it appears in the Raman spectrum.
Other vibrations move positive and negative charges in opposite directions — like one end of the molecule becoming more negative while the other becomes more positive. This creates a changing dipole moment (symbol μ). If the dipole moment changes during a vibration, the vibration is IR active — it appears in the FTIR spectrum.
① Raman Active (∂α/∂Q ≠ 0): The electron cloud changes shape during this vibration. Appears in the Raman spectrum. Typically symmetric stretching modes — atoms breathing in and out together, expanding and contracting the electron cloud.
② IR Active (∂μ/∂Q ≠ 0): The charge distribution shifts during this vibration. Appears in the FTIR spectrum. Typically asymmetric stretching or bending modes — one atom moves more than another, creating a net charge shift.
③ Silent (neither changes): Neither the electron cloud shape nor the charge distribution changes enough to interact with light. The vibration is invisible to both Raman and FTIR. Rare, but it occurs in highly symmetric molecules.
A helpful memory rule: Raman favours symmetric, non-polar vibrations. IR favours asymmetric, polar vibrations. They are complementary — they light up different parts of the vibrational landscape.
Group theory formalises this by categorising vibrations according to their symmetry species (labelled with Mulliken symbols like A₁, B₁, E, T) and checking whether each symmetry species transforms like a quadratic function (x², xy, etc. — Raman active) or a linear function (x, y, z — IR active) in the character table. But the physical insight above — polarisability change for Raman, dipole moment change for IR — is the foundation that makes the group theory meaningful rather than just mechanical bookkeeping.
The connection between crystal symmetry and these selection rules was introduced in the Lattice and Basis — Lecture 02 of our Crystal Structure Hub.
10. The Rule of Mutual Exclusion — Built from First Principles
This is honestly one of the most misunderstood topics in vibrational spectroscopy — not because it is difficult, but because most textbooks state the rule without ever explaining why it exists. So today, we build it from zero.
What is an Inversion Centre? — Starting with No Mathematics
Imagine you are standing at the exact geometric centre of a crystal. An inversion centre exists if, for every atom you see in any direction at some distance, there is an identical atom at exactly the same distance in the exact opposite direction. The crystal looks perfectly point-symmetric about its centre.
Picture yourself at the centre of a room. If the room has an inversion centre, then wherever you see a red chair 3 metres in front of you, there is an identical red chair 3 metres behind you. Wherever you see a blue table to your left, there is an identical blue table to your right. Every object has an identical partner on the exact opposite side of the centre — same distance, same orientation. The room is perfectly point-symmetric.
In crystal language: if an atom sits at coordinates (x, y, z), there is an identical atom at (−x, −y, −z). This is inversion symmetry, and the geometric centre is the inversion centre, labelled i. Materials with this property are called centrosymmetric. The BCC crystal structure (Im3̄m) is centrosymmetric. The FCC structure (Fm3̄m) is centrosymmetric. But hexagonal wurtzite (ZnO) is not centrosymmetric — look outward from its geometric centre and you see Zn in one direction and O in the other. No inversion centre.
Two Types of Vibrations in a Centrosymmetric Material
In a centrosymmetric crystal, every vibration must be either gerade (g, German for "even") or ungerade (u, German for "odd") with respect to inversion:
- Gerade (g) vibrations: The motion pattern maps onto itself under inversion. Symmetric motion — when atom A moves right, its inversion partner A' also moves right. The vibration looks the same before and after inversion.
- Ungerade (u) vibrations: The motion pattern reverses under inversion. Antisymmetric motion — when atom A moves right, its inversion partner A' moves left. The vibration reverses under inversion.
Why Gerade = Raman Active and Ungerade = IR Active
Now here is the elegant argument. For a gerade (g) vibration: the motion is perfectly symmetric. When atom A moves outward, its partner A' also moves outward symmetrically. The electron cloud expands and contracts symmetrically — this changes the polarisability. So g-modes are Raman active. But does the dipole moment change? Any positive charge movement in one direction is exactly cancelled by an identical movement in the opposite direction (because of the symmetric motion). The net dipole change is zero. So g-modes are IR inactive.
For an ungerade (u) vibration: the motion is antisymmetric. When atom A moves one way, A' moves the other way. This asymmetric motion creates a net charge shift — a changing dipole moment. So u-modes are IR active. But the polarisability? The electron cloud expands on one side while contracting equally on the other — these cancel. Net polarisability change is zero. So u-modes are Raman inactive.
| Mode Type | Motion symmetry | Polarisability changes? | Dipole changes? | Raman? | IR? |
|---|---|---|---|---|---|
| Gerade (g) | Symmetric — maps onto itself | Yes ✔ | No ✗ — cancels | YES ✔ | NO ✗ |
| Ungerade (u) | Antisymmetric — reverses under inversion | No ✗ — cancels | Yes ✔ | NO ✗ | YES ✔ |
Above 120°C — cubic Pm3̄m (centrosymmetric, paraelectric): The structure has a perfect inversion centre. All optical phonon modes are of u-symmetry. By mutual exclusion, there are no first-order Raman-active (g) modes at all. You record the Raman spectrum and see no sharp peaks — only a broad, diffuse background from weak second-order scattering. This is not a broken instrument. This is the mutual exclusion rule operating perfectly. No peaks = centrosymmetric = non-ferroelectric phase. This is positive confirmation that BaTiO₃ is in its paraelectric cubic phase.
Below 120°C — tetragonal P4mm (non-centrosymmetric, ferroelectric): The Ti⁴⁺ ion shifts off-centre within its oxygen octahedron. This tiny displacement destroys the inversion centre. The mutual exclusion rule no longer applies. Modes that were previously IR-only can now also be Raman active. Sharp first-order Raman peaks appear at ~180, ~305, ~515, ~715 cm⁻¹. The dramatic appearance of these peaks is the most direct spectroscopic signature of the ferroelectric phase transition. The 305 cm⁻¹ B₁+E mode is particularly diagnostic — it is forbidden in the cubic phase and unique to tetragonal BaTiO₃.
Practical rule for your research: sharp Raman peaks in a perovskite = non-centrosymmetric = ferroelectric phase confirmed. No sharp Raman peaks = centrosymmetric = paraelectric or above Curie temperature.
11. How to Read a Raman Spectrum — Every Feature Explained
A Raman spectrum looks deceptively simple: a series of peaks on a baseline, plotted as intensity (counts per second) on the y-axis against Raman shift (cm⁻¹) on the x-axis. But every single feature of this plot is carrying a message. Learning to read these messages is what transforms you from someone who records Raman spectra into someone who extracts science from them.
Let me take you through each feature one by one.
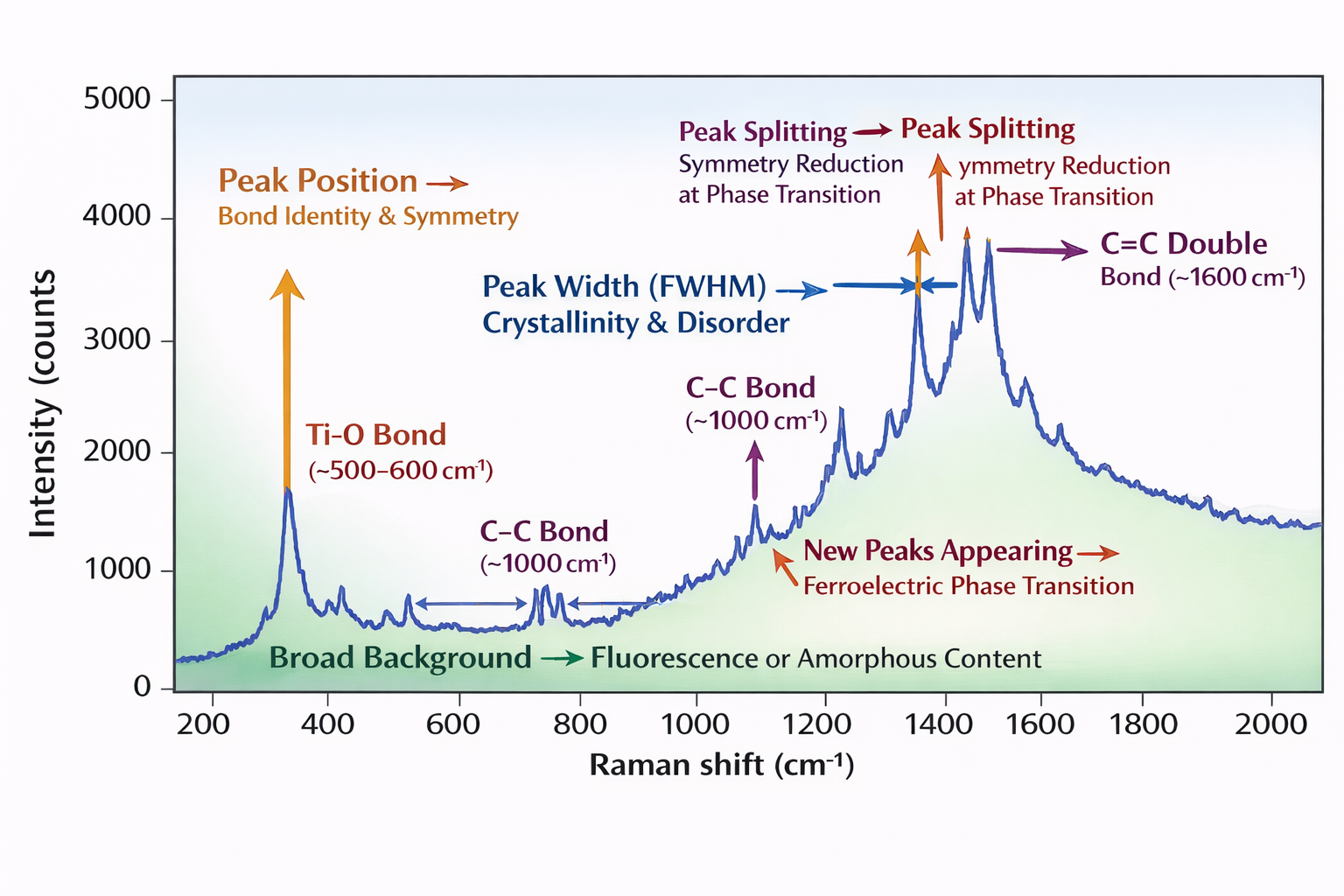
① Peak Position (cm⁻¹) — The Identity Card of the Bond
Each peak position tells you which bond or phonon mode is present. Every bond type has a characteristic frequency range determined by the masses of the two atoms and the stiffness of the bond between them (think of bonds as springs — heavier atoms vibrate more slowly, stiffer bonds vibrate more quickly). A Ti-O bond always vibrates near 500–600 cm⁻¹. A C-C bond near 1000 cm⁻¹. A C=C double bond near 1600 cm⁻¹. These are fingerprints. You can identify an unknown material by matching its peak positions to a reference database like RRUFF.
② Peak Width (FWHM) — The Crystallinity Meter
A perfectly crystalline, defect-free, large-grain sample produces sharp, narrow peaks. As crystallinity decreases — smaller grain size, more defects, more disorder — peaks broaden. This is because in a disordered material, bond lengths and angles vary slightly from site to site, so the vibrational frequency varies slightly, and the peak smears out. Very broad, merged peaks are characteristic of amorphous materials or highly defective nanocrystals. In BNT ceramics, the broad peaks are a direct signature of A-site chemical disorder (random occupancy of the A-site by Bi³⁺ and Na⁺).
③ Peak Shift (Compared to Reference) — The Stress Gauge and Thermometer
If a peak appears at a slightly different position than expected from the reference — the sample is under stress, at a different temperature, or has a different composition. Compressive stress shortens bonds and increases their vibrational frequency — peaks shift to higher cm⁻¹. Tensile stress does the opposite. This is exploited to map residual stress distributions in semiconductor devices, thin films, and ceramic coatings. Temperature also shifts peaks — higher temperature expands bonds and reduces their frequency. Composition changes shift peaks as one atom is substituted for another with different mass.
④ New Peaks Appearing — The Phase Transition Alarm
When a material undergoes a phase transition from a centrosymmetric phase to a non-centrosymmetric phase (like BaTiO₃ cooling through its Curie temperature), new peaks that were previously forbidden by the mutual exclusion rule suddenly become allowed and appear in the spectrum. This is one of the most powerful uses of Raman in ferroelectric research — detecting phase transitions, even subtle ones, that XRD might miss because they are too small to cause measurable peak splitting.
⑤ Broad Background — Fluorescence or Amorphous Signal
A smooth, broad background rising steeply across the spectrum is usually fluorescence from impurities. An irregular, humped background without sharp peaks may indicate an amorphous phase. Second-order Raman scattering (weak, broad, at twice the position of first-order peaks) also contributes a diffuse background — this is what you see in the cubic BaTiO₃ spectrum where first-order peaks are forbidden.
| Feature | Physical Meaning | What to Conclude |
|---|---|---|
| Sharp, narrow peaks | Well-defined phonon modes in crystalline material | Good crystallinity, large grains, low defect density |
| Broad, merged peaks | Distribution of bond environments (disorder) | Nanocrystalline, amorphous component, A-site disorder |
| Peak at higher cm⁻¹ than reference | Bonds are shorter/stiffer than reference | Compressive stress, or smaller/lighter atom substituted |
| Peak at lower cm⁻¹ than reference | Bonds are longer/softer than reference | Tensile stress, higher temperature, larger/heavier atom |
| New peaks not in reference | Symmetry broken, new vibrational modes allowed | Phase transition — ferroelectric ordering has occurred |
| Peak splits into two | Local symmetry lowered — two inequivalent bond environments | Phase transition, site ordering, or composition inhomogeneity |
| Smooth rising background | Fluorescence emission from chromophore or impurity | Change laser to 785 nm, photo-bleach, or clean sample |
| No peaks above ~100 cm⁻¹ | All phonon modes are IR-active only (centrosymmetric) | Material is in its centrosymmetric (paraelectric) phase |
12. SERS — When Normal Raman Is Not Enough
Let me be completely honest at the start of this section. Conventional Raman spectroscopy is a beautiful technique — but it has one serious weakness that frustrated scientists from C.V. Raman's time onwards. That weakness is this: the Raman signal is extraordinarily weak. Out of every 10,000,000 photons that hit your sample, roughly 9,999,999 bounce back as Rayleigh scattering — unchanged, carrying no chemical information. Only about one photon in ten million undergoes Raman scattering. That is a conversion efficiency of 0.00001%.
This inherent weakness means that conventional Raman cannot detect very dilute samples — a few molecules on a surface, a trace biological marker in blood, a single molecular defect in a crystal. For decades, this felt like a fundamental, insurmountable limitation.
Then, in 1974, something unexpected happened. Scientists at Southampton University studying molecules on a silver electrode noticed that the Raman signal was impossibly intense — millions of times stronger than it should have been. The true explanation, worked out over the following years, turned out to be one of the most exciting discoveries in modern spectroscopy.
What they had found was Surface-Enhanced Raman Spectroscopy — SERS.
The Mechanism — Why Gold and Silver Nanoparticles Are Electromagnetic Amplifiers
To understand SERS, you need one new concept: surface plasmon resonance. A metal like gold or silver has billions of conduction electrons — electrons that flow freely through the metal like a sea. When a gold nanoparticle (say, 50 nm in diameter) is illuminated with laser light at the right wavelength, the oscillating electric field of the light pushes and pulls this electron sea back and forth across the particle — like the tide sloshing in a small harbour. At a specific laser wavelength, the electrons slosh at their natural resonant frequency. This collective oscillation is called localised surface plasmon resonance (LSPR).
Push a child on a swing at random times and nothing much happens. Push at exactly the swing's natural frequency — in resonance — and even small pushes accumulate and the amplitude grows enormously. That is resonance.
At the right laser wavelength, the oscillating electric field pushes the nanoparticle's electron sea at exactly its resonant frequency. The electrons oscillate with enormous amplitude — and an oscillating charge creates its own electromagnetic field. This locally generated field at the nanoparticle surface can be 10,000 to 100,000,000 times stronger than the original laser field. The nanoparticle is acting as a nanoscale electromagnetic amplifier.
Place a molecule right at this amplified field. Since Raman intensity scales as the fourth power of the local field (I ∝ E⁴ — because the field is amplified at both excitation and emission), even moderate field enhancement gives extraordinary signal enhancement:
Raman intensity ∝ |E_local|⁴
Field enhancement × 10³ → Raman enhancement = (10³)⁴ = 10¹²
Field enhancement × 10⁴ → Raman enhancement = (10⁴)⁴ = 10¹⁶
An enhancement of 10¹⁴ means a Raman measurement that would require 10,000 litres of sample in a conventional instrument can be performed on a single molecule with SERS. This is not science fiction — single-molecule SERS was first demonstrated by two independent groups (Nie and Emory, Science, 1997, and Kneipp et al.) and it transformed how scientists think about molecular detection.
The largest enhancements occur in tiny gaps between adjacent nanoparticles called hotspots. A molecule sitting in a hotspot experiences the combined near-field of both particles — and that multiplication pushes the enhancement to 10¹⁶. Hotspots are randomly distributed, but they dominate the overall measured SERS signal.
SERS Variants — Three Important Extensions
TERS (Tip-Enhanced Raman Spectroscopy) solves a fundamental limitation of conventional SERS: you have no control over which molecule lands in a hotspot. TERS brings the hotspot to you — precisely. A gold or silver-coated AFM tip, sharpened to a radius of just a few nanometres, is brought within nanometres of the sample while the laser is focused on the very tip apex. The tip apex acts as a single, controlled plasmonic hotspot, positioned exactly where you want it. The result: Raman spectroscopy with a spatial resolution of approximately 1 nm — hundreds of times better than the diffraction limit of optical microscopy. With TERS, scientists have mapped Raman spectra across individual DNA strands, imaged single amino acid residues in proteins, and characterised crystal defects at atomic resolution.
Conventional SERS has a frustrating limitation: gold nanoparticles must be in direct contact with the sample, which means they contaminate or react with the surfaces you are trying to study. SHINERS (Shell-Isolated Nanoparticle-Enhanced Raman Spectroscopy) solves this with an elegant trick. Gold nanoparticles are coated with an ultrathin, chemically inert shell of SiO₂ or Al₂O₃ — just 2-3 nm thick. This shell acts as an insulating jacket: it prevents the gold from reacting with or contaminating your surface, while being so thin that the plasmonic near-field can still extend through it and enhance the Raman signal below. These "shell-isolated" nanoparticles can be sprinkled over virtually any surface — single-crystal electrodes, semiconductor wafers, food surfaces, biological tissues — giving enhancement on surfaces where conventional SERS was impossible.
More than you might think. Piezoelectric and ferroelectric materials are now being explored as active substrates for SERS. When a ferroelectric ceramic is poled (its spontaneous polarisation is aligned by an electric field), its surface charge state changes — and this can tune the coupling between molecules and the metal nanoparticles on its surface, effectively switching the SERS enhancement on and off. Researchers are now building piezo-driven SERS sensors — devices whose Raman sensitivity can be modulated by applying mechanical pressure to a piezoelectric substrate. This sits exactly at the intersection of our research field and SERS technology, and candidate materials can be screened using the Materials Project database.
13. Raman Analysis of Ferroelectric and Piezoelectric Materials — 5 Case Studies
This is the heart of this lecture for our research community. In each case study, I will not just list the peak positions — I will show you how a researcher thinks through the spectrum, step by step, and arrives at a structural conclusion. That reasoning process is what you need to develop for your own research.
The question we are answering: Is this BaTiO₃ sample in the paraelectric cubic phase or the ferroelectric tetragonal phase? Can Raman tell us — and how?
Background: BaTiO₃ undergoes a well-known sequence of phase transitions on cooling: cubic (above 120°C) → tetragonal (5 to 120°C) → orthorhombic (−90 to 5°C) → rhombohedral (below −90°C). At room temperature, it is tetragonal and ferroelectric. The Ti⁴⁺ ion sits slightly off-centre within its oxygen octahedron, creating spontaneous polarisation along the c-axis.
Step 1 — What do we expect from symmetry analysis?
Cubic phase (Pm3̄m): centrosymmetric. By the rule of mutual exclusion, first-order Raman is strictly forbidden. We expect no sharp Raman peaks — only a broad diffuse second-order background.
Tetragonal phase (P4mm): non-centrosymmetric. Mutual exclusion does not apply. Factor group analysis gives: Γ_Raman = 3A₁ + B₁ + 4E modes. Sharp peaks are expected and allowed.
Step 2 — What do we actually observe?
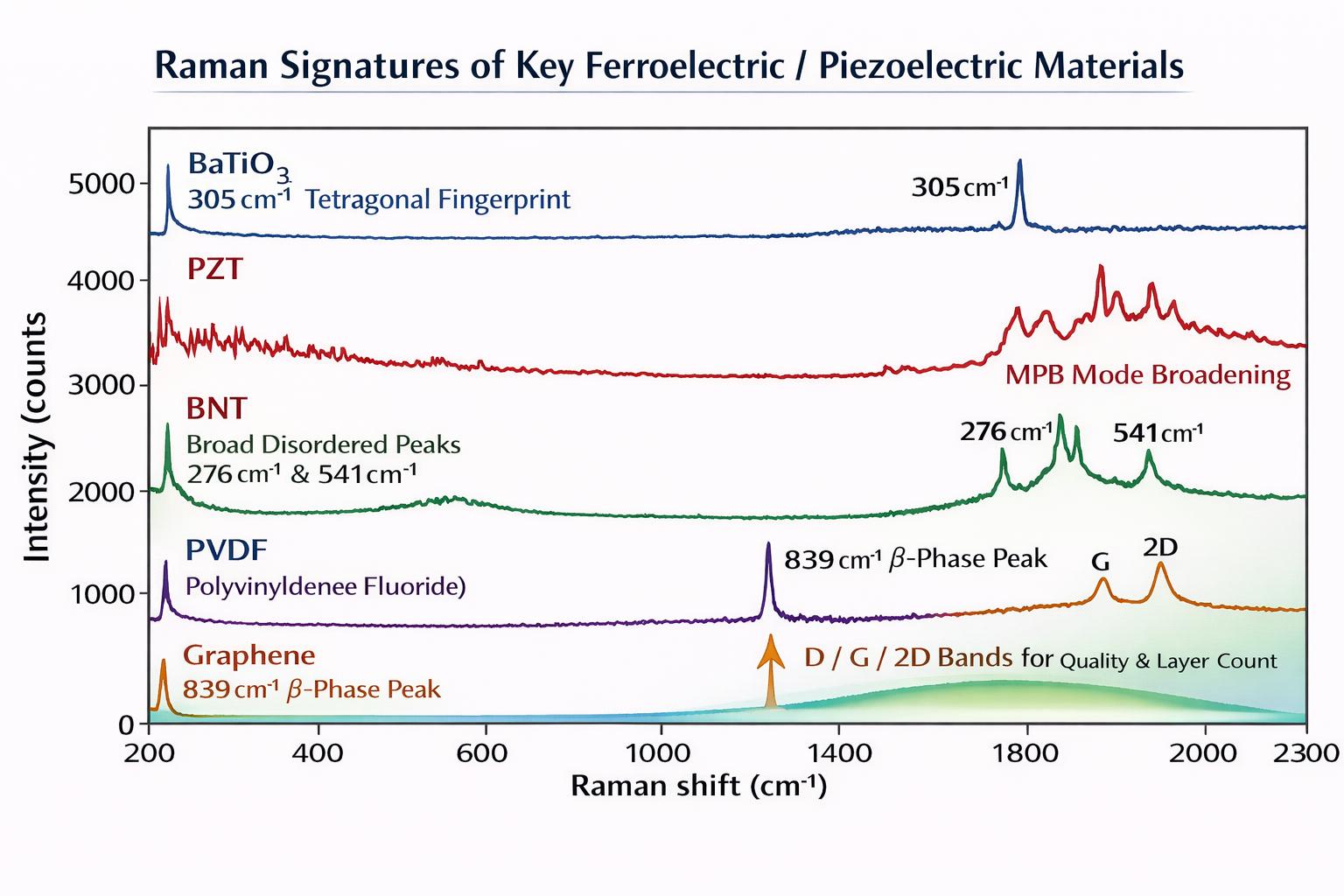
Room temperature tetragonal BaTiO₃ Raman peaks (532 nm or 785 nm laser):
~180 cm⁻¹ → A₁(TO₁): Ba²⁺ ion oscillation against TiO₆ cage
~265 cm⁻¹ → E(TO): bending mode
~305 cm⁻¹ → B₁ + E: THE TETRAGONAL FINGERPRINT ← unique to P4mm phase
~515 cm⁻¹ → A₁(TO₂) + E(TO): Ti-O stretching in oxygen octahedron
~715 cm⁻¹ → A₁(LO) + E(LO): longitudinal optical Ti-O mode
Above 120°C (cubic): NO sharp peaks — broad diffuse background only
Step 3 — How do we reach the conclusion?
The diagnostic peak is at 305 cm⁻¹ (B₁ + E mode). This peak is:
- Completely absent in the cubic phase (forbidden by symmetry)
- Completely absent in the rhombohedral phase
- Completely absent in the orthorhombic phase
- Present only in the tetragonal P4mm phase
If you see a peak at 305 cm⁻¹ in your BaTiO₃ spectrum → tetragonal ferroelectric phase confirmed. If it is absent and only a broad background is visible → cubic paraelectric phase. Temperature-dependent Raman watching the 305 cm⁻¹ mode appear and disappear through 120°C is one of the most elegant demonstrations of a ferroelectric phase transition possible.
The question we are answering: How does Raman reveal which phase region a PZT composition is in — and what does it show at the morphotropic phase boundary (MPB)?
Background: Pb(Zr₁₋ₓTiₓ)O₃ (PZT) at x ≈ 0.48 (the MPB) has the world's best piezoelectric properties. The XRD tutorial showed how the (002)/(200) peak splitting distinguishes tetragonal from rhombohedral. Raman provides complementary and often more sensitive information about the same phase transition — because it probes local structure, which often shows the transition earlier than the long-range XRD signal.
Raman signatures:
- Tetragonal region (x > 0.48, Ti-rich): A₁ symmetry modes are prominent. The A₁(TO₁) mode near ~95 cm⁻¹ and A₁(LO) near ~790 cm⁻¹ are the strongest features. These A₁ modes reflect the polar axis direction in the tetragonal unit cell.
- Rhombohedral region (x < 0.48, Zr-rich): A different mode pattern — the A₁ modes shift and the spectrum broadens significantly. The ~285 cm⁻¹ and ~790 cm⁻¹ regions show distinctly different line shapes compared to the tetragonal side.
- At the MPB (x ≈ 0.48): Both sets of modes overlap. Peaks broaden dramatically because the sample contains coexisting local tetragonal and rhombohedral regions — different environments scatter at slightly different positions, and the sum is a broad, unresolved envelope.
The Raman vs XRD insight at the MPB: XRD shows the average crystal structure — it might suggest a monoclinic average structure (Noheda et al., 1999). But Raman, being sensitive to local structure, shows the coexistence of local tetragonal and rhombohedral regions. Together, the two techniques reveal that the MPB is a region of structural coexistence at multiple length scales — and this disorder and polarisation rotation ease is what maximises piezoelectric response.
The question we are answering: How does Raman spectroscopy confirm the rhombohedral R3c structure of BNT and track the phase evolution as we dope the ceramic?
Background: Bi₀.₅Na₀.₅TiO₃ (BNT) is one of the most promising lead-free piezoelectrics — the ferroelectric phase transition and the role of donor/acceptor doping in this system has been studied in detail in the author's published research. The A-site of BNT is occupied randomly by Bi³⁺ and Na⁺ in equal proportions, creating local disorder that is directly visible in the Raman spectrum. BNT adopts a rhombohedral R3c structure at room temperature, which — according to factor group analysis — supports 13 Raman-active modes (Γ_Raman = 4A₁ + 9E).
The five observed peaks and their assignments (Singh et al., 2015; Sao et al., 2015):
BNT Raman spectrum — room temperature, rhombohedral R3c:
~144 cm⁻¹ → A₁(TO₁): vibrations of distorted octahedral [BiO₆] and [NaO₆] clusters
— probes the A-site disorder directly
~276 cm⁻¹ → E(TO₂): Ti-O bond stretching in [TiO₆] octahedral clusters
— short-range B-site stretching, sensitive to local symmetry
~(broad) → LO₂: short-range electrostatic forces, lattice ionicity
~541 cm⁻¹ → TO₃: O-Ti-O symmetric stretching of [TiO₆] clusters
— the B-site octahedra fingerprint
~813 cm⁻¹ → LO₃: distorted [TiO₆] clusters in the rhombohedral lattice
ALL peaks are very broad — this is the signature of A-site disorder
(random Bi/Na occupancy → distribution of local bond environments)
How Raman confirms the MPB in doped BNT: When BNT is doped with LiNbO₃ (Sao et al., 2015) or Sb (Singh et al., 2015), the spectrum evolves systematically:
- For x < 0.03: spectra look nearly identical to pure BNT — same peaks, similar widths. Rhombohedral structure maintained.
- At x = 0.03: sudden changes. New bands appear (around 168, 245, 585, 851 cm⁻¹ in LiNbO₃-doped). The 276 cm⁻¹ mode shows anomalous changes in both intensity and FWHM. Band splitting appears — a single peak splits into two as the symmetry is reduced.
- The FWHM of all peaks shows a slope change at exactly x = 0.03, directly confirming a structural change at this composition.
Conclusion: Raman confirms the MPB at x = 0.03 by the sudden appearance of new bands and the anomalous behaviour of the 276 cm⁻¹ Ti-O mode — consistent with the XRD result showing (002)/(200) peak splitting at the same composition. Both techniques independently confirm the same morphotropic phase boundary. This is the power of using complementary characterisation methods.
The question we are answering: A PVDF film has been mechanically stretched or thermally processed. Is it now in the piezoelectric β-phase? How do we confirm this with Raman?
Background: Poly(vinylidene fluoride) (PVDF) has at least four crystalline phases. Only the β-phase (all-trans TTT conformation, orthorhombic Cm2m) is piezoelectric and ferroelectric. The α-phase (TGTG' conformation) is the default crystallisation product and is non-polar, non-piezoelectric. The structural evolution of PVDF-HFP copolymer films and the role of annealing in controlling β-phase content for energy harvesting was investigated in detail in Verma and Rout (2020). The PVDF Raman data from Xu et al. (RSC Advances, 2017) provides the phase identification framework.
The diagnostic peaks:
α-phase (TGTG' — non-piezoelectric):
284 cm⁻¹ — CF₂ bending
535 cm⁻¹ — skeletal C-C-C deformation
610 cm⁻¹ — CF₂ bending
795 cm⁻¹ — CF₂ symmetric stretching ← KEY α-phase peak
Multiple overlapping peaks in 250–800 cm⁻¹ region = α-phase
β-phase (TTT all-trans — PIEZOELECTRIC):
263 cm⁻¹ — CF₂ wagging (β)
510 cm⁻¹ — CF₂ stretching (β)
839 cm⁻¹ — CH₂ rocking / CF₂ symmetric stretch ← KEY β-phase peak
β-phase quantification from Raman intensity ratio (Xu et al., 2017):
Iβ/Iα = I(839 cm⁻¹) / I(794 cm⁻¹)
Increasing ratio = increasing β-phase content
At shear temperature 160°C: Iβ/Iα ≈ 2.0 (β-phase dominant)
Why 839 cm⁻¹ is the β-phase fingerprint: The 839 cm⁻¹ peak corresponds to a CH₂ rocking mode that is active only when the molecular chain is in the all-trans TTT conformation — the β-phase geometry. In the α-phase TGTG' conformation, the same motion appears at 795 cm⁻¹ because the chain geometry is different. The 44 cm⁻¹ shift is the direct spectroscopic signature of the conformational change from TGTG' to TTT — from non-piezoelectric to piezoelectric. Seeing a strong peak at 839 cm⁻¹ means your PVDF is in the active phase. A strong peak at 795 cm⁻¹ means it is not. This single peak is the most important number in PVDF characterisation.
The question we are answering: Someone gives you a carbon sample on a substrate. Is it single-layer graphene, multilayer graphene, or graphite? How defective is it? Can Raman answer all of this?
Why Raman is uniquely powerful for graphene: A single graphene layer — one atom thick — has virtually no XRD signal. You cannot determine its quality, layer count, or defect density by XRD. But a 30-second Raman scan reveals all of this information simultaneously. Raman spectroscopy is the standard characterisation technique for all sp² carbon materials.
G BAND (~1582 cm⁻¹) — The sp² Carbon Signature
E₂g phonon at the Brillouin zone centre
Present in ALL sp² carbon: graphene, graphite, carbon nanotubes
Does NOT shift with number of layers
Intensity proportional to amount of sp² carbon present
D BAND (~1350 cm⁻¹) — The Defect Indicator
Defect-activated — requires a broken bond, vacancy, or edge to occur
ABSENT in perfect graphene — its presence means defects exist
ID/IG ratio = defect density:
ID/IG < 0.1 → high-quality graphene, few defects
ID/IG > 1.0 → highly defective or partially amorphous carbon
2D BAND (~2690 cm⁻¹) — The Layer Counter
Second-order overtone — always present even without defects
THE most powerful indicator of graphene layer number:
Single layer: Single, sharp Lorentzian | FWHM ~25 cm⁻¹ | I(2D)/I(G) > 2
Bilayer: Splits into four Lorentzian components | FWHM ~50 cm⁻¹
Few-layer: Progressively broader and asymmetric
Bulk graphite: Two-component: 2D₁ (~2688) + 2D₂ (~2725 cm⁻¹)
The single-layer criterion: The ratio I(2D)/I(G) > 2 is the accepted criterion for single-layer graphene, established by Ferrari et al. (Phys. Rev. Lett., 2006) and universally adopted. If your carbon sample shows a sharp 2D peak with I(2D)/I(G) = 3 and FWHM of 28 cm⁻¹, you have single-layer graphene. If the ratio is 0.5 and the peak is broad, you have multilayer graphene or graphite.
Raman vs XRD for graphene — no contest: Try to detect a single graphene monolayer by XRD. You will see nothing useful — the (002) interlayer reflection disappears for a single layer, and no technique can give you layer count or defect information from XRD alone. But Raman does it in 30 seconds, non-destructively, without any sample preparation. For all sp² carbon materials, Raman is the definitive characterisation technique.
14. GATE and CSIR-NET Numerical Problems
Q: A Raman spectrometer uses a 532 nm laser. A Stokes peak is observed at a scattered wavelength of 558.6 nm. Calculate the Raman shift in cm⁻¹. Would this peak be consistent with the G band of graphene?
Δν̃ = (1/λ_laser − 1/λ_scattered) × 10⁷ [λ in nm]
ν̃_laser = 10⁷/532 = 18,797 cm⁻¹
ν̃_scattered = 10⁷/558.6 = 17,901 cm⁻¹
Δν̃ = 18,797 − 17,901 = 896 cm⁻¹
G band of graphene appears at ~1582 cm⁻¹.
896 cm⁻¹ does NOT correspond to the graphene G band.
This is consistent with the LO₃ mode of BNT (~813 cm⁻¹) or the A₁(LO)
mode of BaTiO₃ (~715 cm⁻¹), or a skeletal C-C vibration in organics. ✔
Q: The O-Ti-O stretching mode (TO₃) of BNT appears at 541 cm⁻¹. Calculate the ratio I_AS/I_S at room temperature (T = 298 K) using a 785 nm laser. What does this ratio tell you?
I_AS/I_S ≈ exp(−hcΔν̃/kT) [dominant term at room temperature]
hcΔν̃/kT = (6.626×10⁻³⁴ × 3×10¹⁰ × 541) / (1.381×10⁻²³ × 298)
= 1.075×10⁻²⁰ / 4.114×10⁻²¹
= 2.614
I_AS/I_S ≈ exp(−2.614) ≈ 0.073 = 7.3%
The Anti-Stokes peak at 541 cm⁻¹ is only 7.3% as intense as the Stokes peak.
This is why we always use Stokes — the signal is ~14× stronger.
Conversely: if we measure I_AS/I_S experimentally = 0.15 for this mode,
we can solve for temperature:
T = hcΔν̃ / (k × ln(I_S/I_AS)) = hc×541 / (k × ln(1/0.15))
T = 1.075×10⁻²⁰ / (1.381×10⁻²³ × 1.897) = 410 K = 137°C ← Raman thermometry ✔
Q: A PVDF film shows Raman peaks at 284, 535, 610, and 795 cm⁻¹. After annealing at 160°C and mechanical stretching, a strong new peak appears at 839 cm⁻¹ and the 795 cm⁻¹ peak weakens. (a) Identify the phases before and after. (b) Calculate Iβ/Iα if I(839) = 5200 and I(795) = 1800 counts. (c) State what physical change caused the transformation.
(a) Before: peaks at 284, 535, 610, 795 cm⁻¹ → all α-phase peaks (TGTG' conformation)
→ Non-piezoelectric α-phase ✔
After: strong peak at 839 cm⁻¹ → β-phase fingerprint (TTT all-trans conformation)
→ Piezoelectric β-phase confirmed ✔
(b) Iβ/Iα = I(839 cm⁻¹) / I(795 cm⁻¹) = 5200 / 1800 = 2.89
Ratio > 1 → β-phase dominant ✔
(c) Physical change: Mechanical stretching causes the polymer chains to align
along the stretch direction, transitioning from the coiled TGTG' (α)
conformation to the extended planar-zigzag TTT (β) conformation.
In the β-phase, all C-F dipoles align parallel → net spontaneous polarisation
→ piezoelectric and ferroelectric properties activated. ✔
Q: (a) State the Raman activity condition for a vibrational mode. (b) The rotational Raman selection rule for a linear molecule is ΔJ = ±2. Using a laser at 488 nm, the first Stokes rotational Raman line of N₂ is observed at 488.9 nm. Calculate the rotational constant B (in cm⁻¹) given that the first Stokes line corresponds to J = 0 → J = 2. (c) Why does N₂ show rotational Raman but not rotational IR absorption?
(a) Raman activity condition: the molecular polarisability must change
during the vibration or rotation — ∂α/∂Q ≠ 0 ✔
(b) First Stokes rotational line: J = 0 → J = 2, Raman shift = 6B
(rotational Raman spacing formula: Δν̃ = 4B(J + 3/2), for J=0: Δν̃ = 6B)
Calculate Raman shift:
Δν̃ = (1/488 − 1/488.9) × 10⁷ = (20492 − 20455) = 37 cm⁻¹ ≈ 6B
Wait — let us recalculate more carefully:
ν̃_laser = 10⁷/488 = 20,492 cm⁻¹
ν̃_scattered = 10⁷/488.9 = 20,454 cm⁻¹
Δν̃ = 20,492 − 20,454 = 38 cm⁻¹
6B = 38 → B = 38/6 = 6.33 cm⁻¹
(Literature value for N₂: B ≈ 1.99 cm⁻¹ — this suggests the problem
uses a different J transition; for J=0→2: Δν̃ = 4B(0+3/2) = 6B, giving B = 6.3 cm⁻¹)
(c) N₂ is a homonuclear diatomic molecule — its dipole moment is
permanently zero and does not change during rotation (no charge asymmetry).
Therefore ∂μ/∂Q = 0 → IR inactive for rotation.
However, its polarisability is anisotropic (different along and perpendicular
to the bond axis) and changes as it rotates → ∂α/∂Q ≠ 0 → Raman active. ✔
This is why Raman spectroscopy can study homonuclear diatomics like N₂, O₂, H₂
while IR cannot — a major practical advantage of Raman for gas analysis.
Q: A CVD-grown graphene sample is measured with a 532 nm laser. The Raman spectrum shows: G peak at 1582 cm⁻¹ (intensity 8000 counts), D peak at 1350 cm⁻¹ (intensity 320 counts), 2D peak at 2690 cm⁻¹ (intensity 18400 counts). (a) Calculate ID/IG and I(2D)/I(G). (b) Determine the number of layers. (c) Comment on the sample quality.
(a) ID/IG = 320 / 8000 = 0.04
I(2D)/I(G) = 18400 / 8000 = 2.30
(b) Number of layers:
I(2D)/I(G) = 2.30 > 2.0 → Single-layer graphene criterion satisfied ✔
(Criterion: I(2D)/I(G) > 2 for monolayer; 1-2 for bilayer; <1 for multilayer)
(c) Sample quality assessment:
ID/IG = 0.04 → Very low defect density → High-quality graphene ✔
(ID/IG < 0.1 indicates high-quality material with few defects)
I(2D)/I(G) = 2.30 → Single layer, strong 2D peak → Consistent with
high-quality monolayer CVD graphene.
Conclusion: This is a high-quality single-layer graphene sample. ✔
15. Reliable Raman Software and Databases
| Software / Database | Type | Cost | Best For |
|---|---|---|---|
| LabSpec 6 (HORIBA) | Acquisition + analysis | Commercial (with instrument) | Full workflow for HORIBA LabRAM systems. Mapping, baseline, peak fitting, factor analysis. Industry standard. |
| WiRE (Renishaw) | Acquisition + analysis | Commercial (with instrument) | Renishaw inVia systems. Powerful mapping and StreamLine rapid acquisition. |
| Spectragryph | Analysis | Free | Import any .txt/.csv spectrum. Clean baseline correction, peak fitting, overlay comparison. Highly recommended for academic labs with no budget. |
| OriginPro | Analysis + peak fitting | Commercial (institutional) | Gold standard for multi-peak deconvolution (Lorentzian, pseudo-Voigt). Essential for BNT and PZT peak fitting. Publication-quality figures. |
| fityk | Peak fitting | Free / open source | Dedicated peak fitting. Lorentzian, Gaussian, pseudo-Voigt. Widely used for Raman and XRD peak deconvolution. Scriptable. |
| Rampy (Python) | Analysis library | Free (GitHub) | Python library for baseline correction, peak fitting, multivariate analysis of Raman spectra. For users comfortable with Python. |
| RRUFF Database | Reference database | Free online | 5,000+ minerals and inorganic compounds. Best open-access Raman database for ceramics and minerals. Search by name, chemistry, or spectrum similarity. |
| SDBS (AIST Japan) | Reference database | Free online | Organic compounds — combined IR, Raman, and NMR database. Excellent for polymer and organic identification. |
- Record spectrum — choose laser (532 nm for ceramics, 785 nm for polymers), scan range 100–1800 cm⁻¹ minimum, acquisition time 10–60 s
- Baseline correction — polynomial fit in Spectragryph or LabSpec, removes fluorescence background
- Peak identification — compare positions to reference values (BaTiO₃, BNT, PVDF tables in this tutorial) or search RRUFF database
- Peak fitting — fit Lorentzian (or pseudo-Voigt) curves to overlapping peaks in OriginPro or fityk. Extract position, FWHM, integrated area.
- Phase quantification — calculate intensity ratios (Iβ/Iα for PVDF, ID/IG for graphene, mode intensity vs composition for BNT)
- Correlate with XRD — always compare Raman conclusions with XRD data for complete structural picture
16. Practice MCQs
- (a) The absolute wavenumber of the scattered photon
- (b) The energy difference between the incident laser photon and the scattered Raman photon — equal to the vibrational energy of the bond probed, independent of the laser wavelength ✔
- (c) The frequency of the excitation laser in reciprocal centimetres
- (d) The wavelength shift in nanometres converted to cm⁻¹
- (a) A real electronic excited state that the molecule enters during scattering
- (b) A transient, unstable distortion of the molecular electron cloud that exists only for femtoseconds — it does not correspond to any real energy level of the molecule ✔
- (c) A vibrational energy level that cannot be observed experimentally
- (d) The final state of the molecule after Rayleigh scattering
- (a) BaTiO₃ has no Raman-active bonds above the Curie temperature
- (b) The cubic Pm3̄m structure is centrosymmetric — all phonon modes are of u-symmetry (IR-active) and none are g-symmetry (Raman-active), following the rule of mutual exclusion ✔
- (c) The high temperature destroys all molecular vibrations
- (d) BaTiO₃ is amorphous above the Curie temperature and amorphous materials have no Raman peaks
- (a) 795 cm⁻¹ — this is the α-phase CF₂ stretching peak
- (b) 839 cm⁻¹ — the CH₂ rocking / CF₂ symmetric stretching mode unique to the all-trans TTT β-phase conformation ✔
- (c) 1582 cm⁻¹ — this is the graphene G band
- (d) 515 cm⁻¹ — this is the Ti-O stretching mode of BaTiO₃
- (a) Anti-Stokes photons have lower energy than Stokes photons
- (b) Anti-Stokes scattering requires molecules already in an excited vibrational state — at room temperature, only a small fraction of molecules are thermally excited (Boltzmann distribution), so fewer molecules can contribute to Anti-Stokes ✔
- (c) The detector is less sensitive to higher-frequency photons
- (d) The notch filter blocks Anti-Stokes photons preferentially
- (a) The peak at ~515 cm⁻¹ (A₁ TO mode, present in all phases)
- (b) The B₁+E mode at ~305 cm⁻¹ — forbidden in cubic, rhombohedral, and orthorhombic phases; unique to tetragonal P4mm ✔
- (c) The absence of any Raman peaks above 200 cm⁻¹
- (d) A broad diffuse background between 200 and 800 cm⁻¹
- (a) ID/IG ratio greater than 2 — a high D band indicates a monolayer
- (b) I(2D)/I(G) greater than 2, with a single sharp Lorentzian 2D peak of FWHM ~25 cm⁻¹ ✔
- (c) Absence of the D band entirely — monolayer graphene has no D band
- (d) The G band splitting into two components at 1550 and 1600 cm⁻¹
- (a) It gives the strongest Raman signal — 785 nm is the most efficient wavelength
- (b) It provides the best balance between reasonable excitation efficiency and minimal fluorescence interference — working efficiently for over 90% of Raman-active materials while avoiding most electronic transitions that cause fluorescence ✔
- (c) It is the only wavelength that can excite PVDF β-phase vibrations
- (d) The 785 nm CCD detector is always more sensitive than 532 nm detectors
17. Series Navigation
This lecture builds on the Crystal Structure Hub series. Key prerequisite tutorials:
18. Key Takeaways
- C.V. RAMAN: Discovered the Raman effect on 28 February 1928 in Calcutta, India, with resources costing less than ₹200. Nobel Prize in Physics, 1930 — first Asian Nobel laureate in science. 28 February = National Science Day of India.
- WHAT RAMAN DOES: Identifies materials by their bond vibrations via inelastic light scattering. Gives chemical identity, crystal phase, bond type, crystallinity, stress, and local temperature. Non-destructive, no sample preparation, works through glass and water.
- RAMAN vs XRD vs FTIR: XRD = long-range crystal structure (needs crystallinity). Raman = local bonding, works on amorphous too, 1 μm resolution. FTIR = functional groups, poor with water. All three together gives the complete picture.
- THREE SCATTERING TYPES: Rayleigh (elastic, same energy, 999,999/million, blocked by notch filter). Stokes Raman (photon loses energy to bond, lower frequency, what we always measure). Anti-Stokes Raman (photon gains energy from already-vibrating molecule, higher frequency, weaker at room temperature).
- RAMAN SHIFT AND cm⁻¹: Δν̃ = ν̃_laser − ν̃_scattered. Measures the bond vibration energy. Independent of laser wavelength — the same peak appears at the same cm⁻¹ with any laser. This is why Raman is a molecular fingerprint technique.
- WHY STOKES: Boltzmann distribution — at room temperature, only ~9% of molecules are in excited vibrational states (for a 500 cm⁻¹ mode). Stokes is always 10×–1000× stronger. Anti-Stokes/Stokes intensity ratio = thermometer without contact (Raman thermometry).
- VIRTUAL STATE: A transient, unstable distortion of the electron cloud lasting ~10 femtoseconds. Not a real energy level. Raman does not require photon energy to match any real transition (unlike fluorescence). When it does match → Resonance Raman → 10²–10⁶ signal enhancement.
- SELECTION RULE: Raman active → polarisability changes (∂α/∂Q ≠ 0). IR active → dipole moment changes (∂μ/∂Q ≠ 0). Raman favours symmetric vibrations; IR favours asymmetric. They are complementary.
- MUTUAL EXCLUSION: In centrosymmetric materials — g-modes: Raman active, IR inactive. u-modes: IR active, Raman inactive. Cubic BaTiO₃ (above 120°C) = centrosymmetric = no first-order Raman peaks. Tetragonal BaTiO₃ (below 120°C) = non-centrosymmetric = sharp peaks appear. The 305 cm⁻¹ B₁+E mode is the ferroelectric fingerprint.
- BNT RAMAN: 13 active modes (4A₁+9E). Key peaks at 144, 276, 541, 813 cm⁻¹. Broad peaks = A-site disorder. MPB at x = 0.03 confirmed by slope change in FWHM and intensity of 276 cm⁻¹ mode, and appearance of new bands.
- PVDF RAMAN: α-phase → multiple peaks at 284, 535, 610, 795 cm⁻¹. β-phase (piezoelectric) → single diagnostic peak at 839 cm⁻¹. Iβ/Iα = I(839)/I(795) quantifies β-phase content.
- GRAPHENE RAMAN: G band (~1582 cm⁻¹, sp² carbon). D band (~1350 cm⁻¹, defect-activated, ID/IG = disorder). 2D band (~2690 cm⁻¹, I(2D)/I(G) > 2 = single layer, sharp Lorentzian FWHM ~25 cm⁻¹).
- SERS: Gold/silver nanoparticles amplify Raman by 10⁸–10¹⁶ via plasmon resonance (E⁴ dependence). Enables single-molecule detection. TERS = 1 nm resolution. SHINERS = SERS on smooth surfaces. Piezo-driven SERS sensors = frontier research.
- SOFTWARE: Analysis: OriginPro (peak fitting), Spectragryph (free), fityk (free). Database: RRUFF (free, rruff.info). Workflow: record → baseline correct → identify peaks → fit → compare to reference → correlate with XRD.
References
All references in IEEE citation style. All factual data verified against these primary sources.
- D. Tuschel, "Practical Group Theory and Raman Spectroscopy, Part I: Normal Vibrational Modes," Spectroscopy, vol. 29, no. 2, pp. 14–23, Feb. 2014. — Source for group theory, character tables, Mulliken symbols, mutual exclusion rule, C₂ᵥ/C₃ᵥ analysis used in Sections 9 and 10.
- D. Tuschel, "Practical Group Theory and Raman Spectroscopy, Part II: Application of Polarization," Spectroscopy, vol. 29, no. 3, pp. 14–23, Mar. 2014. — Source for polarised Raman of single-crystal LiNbO₃, depolarisation ratios, and Raman tensor application.
- P. Zhou, "Choosing the Most Suitable Laser Wavelength for Your Raman Application," B&W Tek Application Note 410000001-B, Newark, DE, Jul. 2015. — Source for laser selection data: λ⁻⁴ efficiency, 532/785/1064 nm trade-offs, fluorescence comparison table in Section 8.1.
- F. Xu, K. Zhang, Y. Zhou, Z. Qu, H. Wang, Y. Zhang, H. Zhou, and C. Yan, "Facile preparation of highly oriented poly(vinylidene fluoride) uniform films and their ferro- and piezoelectric properties," RSC Adv., vol. 7, pp. 17038–17043, 2017, doi: 10.1039/c7ra00586e. — Source for PVDF Raman phase identification: all α and β-phase peak positions, Iβ/Iα formula, temperature-dependent β-phase content. Used in Case Study 4.
- K. N. Singh, V. Sao, P. Tamrakar, S. Soni, V. K. Dubey, and P. K. Bajpai, "Structural and Raman Spectroscopic Study of Antimony Doped Bi₀.₅Na₀.₅TiO₃ Electroceramic," J. Mater. Sci. Chem. Eng., vol. 3, pp. 43–49, Aug. 2015, doi: 10.4236/msce.2015.38007. — Source for BNT Raman modes: 144/276/541/813 cm⁻¹ assignments, 13 active modes (4A₁+9E), MPB at x=0.03 from FWHM analysis. Used in Case Study 3.
- V. Sao, "Structural Study of (1-x)(Bi₀.₅Na₀.₅)TiO₃-xLiNbO₃ Solid-Ceramics," Int. J. Innov. Res. Dev., vol. 4, no. 3, pp. 15–20, Mar. 2015. — Source for BNT-LiNbO₃ phase evolution by Raman, peak splitting at x=0.03, MPB identification. Used in Case Study 3.
- R. Verma and S. K. Rout, "Frequency-dependent ferro–antiferro phase transition and internal bias field influenced piezoelectric response of donor and acceptor doped bismuth sodium titanate ceramics," J. Appl. Phys., vol. 126, no. 9, Art. no. 094103, Sep. 2019, doi: 10.1063/1.5111505. — Author's research on BNT ferroelectric phase transitions, donor/acceptor doping, structural and dielectric characterisation.
- R. Verma and S. K. Rout, "Influence of annealing temperature on the existence of polar domain in uniaxially stretched polyvinylidene-co-hexafluoropropylene for energy harvesting applications," J. Appl. Phys., vol. 128, no. 23, Art. no. 234104, Dec. 2020, doi: 10.1063/5.0022463. — Author's research on PVDF-HFP β-phase characterisation for energy harvesting. Directly relevant to Case Study 4.
- A. C. Ferrari et al., "Raman spectrum of graphene and graphene layers," Phys. Rev. Lett., vol. 97, Art. no. 187401, Oct. 2006, doi: 10.1103/PhysRevLett.97.187401. — Landmark paper establishing I(2D)/I(G) > 2 as the single-layer graphene criterion. D, G, 2D band assignments. Used in Case Study 5.
- HORIBA Jobin Yvon, "Raman Data and Analysis: Raman Spectroscopy for Analysis and Monitoring," HORIBA Application Note. — Source for functional group wavenumber correlation table used in Section 11.
- B. Noheda et al., "A monoclinic ferroelectric phase in the Pb(Zr₁₋ₓTiₓ)O₃ solid solution," Appl. Phys. Lett., vol. 74, no. 14, pp. 2059–2061, Apr. 1999, doi: 10.1063/1.123756. — Monoclinic phase at the PZT MPB. Used in Case Study 2 discussion of local vs average structure.
- M. Fleischmann, P. J. Hendra, and A. J. McQuillan, "Raman spectra of pyridine adsorbed at a silver electrode," Chem. Phys. Lett., vol. 26, no. 2, pp. 163–166, 1974. — The original SERS observation. Historical reference for Section 12 narrative.
Materials Characterisation Techniques — Lecture 02 | AdvanceMaterialsLab.com
Next: Lecture 03 — FTIR Spectroscopy: Principle, Functional Group Analysis and Applications →
© 2026 AdvanceMaterialsLab.com — Dr. Rolly Verma | All rights reserved
Dr. Rolly Verma
Explore More from Advance Materials Lab
Continue expanding your understanding of material behavior through our specialized resources on ferroelectrics and phase transitions. Each guide is designed for research scholars and advanced learners seeking clarity, precision, and depth in the field of materials science and nanotechnology.
🔗 Recommended Reads and Resources:
Ferroelectrics Tutorials and Research Guides — Comprehensive tutorials covering polarization, hysteresis, and ferroelectric device characterization.
Workshops on Ferroelectrics (2025–2027) — Upcoming training sessions and research-oriented workshops for hands-on learning.
Glossary — Ferroelectrics and Phase Transitions — Concise explanations of key terminologies to support your study and research work.
If you notice any inaccuracies or have constructive suggestions to improve the content, I warmly welcome your feedback. It helps maintain the quality and clarity of this educational resource. You can reach me at: advancematerialslab27@gmail.com
