Crystal Structure
Raman Spectroscopy
Research Careers
Ferroelectrics & Testing Systems
Introduction to Crystal Structure
Why the Arrangement of Atoms Governs the Physical World By Dr. Rolly Verma | AdvanceMaterialsLab.com | March 2026 | B.Sc. / M.Sc. Materials Science & EngineeringSeries: Crystal Structure Hub | Lecture: 01 of 10 | Level: Beginner — No prior materials science knowledge required
Reading time: 25 minutes | Includes: Core concepts, carbon analogy, band theory introduction, functional materials, 2025 frontiers, MCQs, key takeaways
SEO Keywords: introduction to crystal structure, why crystal structure is important, crystal structure materials science, crystalline vs amorphous, atomic arrangement properties, crystal structure applications
1. Introduction: The Question at the Heart of Materials Science
Welcome to the Crystal Structure Hub. This is the first lecture in a series designed to take you from the very fundamentals of atomic arrangement all the way to the mathematical and physical framework that underpins modern materials science, nanotechnology, and quantum device engineering.
Before we study semiconductors, before we discuss ferroelectrics, piezoelectrics, or topological quantum materials, we must answer a question that is deceptively simple but profoundly important:
"If we already know what atoms a material contains, why should we care about how those atoms are arranged?"
The answer is the foundation of everything in this course: the properties of a material are determined not only by what atoms it contains, but critically by how those atoms are arranged in space. This principle is not a philosophical claim — it is experimentally verified, technologically validated, and mathematically grounded in solid-state physics.
This single principle explains why diamonds are harder than graphite, why silicon can power your phone, why some materials generate electricity when you press them, and why a new class of quantum materials discovered in 2025 could one day enable dissipationless electronics. In each case, the atomic arrangement — the crystal structure — is the governing factor.
2. What is a Crystal? Defining Order in Solids
In materials science, a crystal is defined not by its external shape, but by its internal order. You may have seen beautiful gemstones with flat faces and sharp edges, but that external form is merely a consequence of something far more fundamental — the periodic arrangement of atoms inside.
A crystalline solid is one in which atoms are arranged in a periodic, repeating pattern that extends uniformly in three dimensions throughout the material. This repeating pattern is described mathematically as a lattice with an associated basis — a concept we will study rigorously in the next lecture.
Imagine a floor tiled with identical square tiles in a perfectly regular pattern. If you stand anywhere on the floor and look around, you see exactly the same arrangement of tiles in every direction. That regularity — that periodicity — is what makes it a crystal. Now imagine the same floor made of irregularly shaped, randomly sized pieces with no repeating pattern. That is an amorphous solid. Same material, completely different behaviour.
2.1 Periodicity: The Key Concept
What makes crystals so special from a physics perspective is this periodicity. When atoms repeat in a regular, ordered fashion, they create symmetry. And symmetry is not merely geometric beauty — it is a physical constraint that governs how a material responds to mechanical stress, electric fields, magnetic fields, heat, and light.
The branch of science that formally studies this symmetry is called crystallography. As classified by the International Union of Crystallography (IUCr), all possible crystal symmetries in three dimensions are grouped into 7 crystal systems, 14 Bravais lattices, and 230 space groups. These mathematical structures are the complete catalogue of all ways that atoms can arrange themselves periodically in three-dimensional space.
2.2 Crystalline vs Amorphous — Why It Matters
The contrast between crystalline and amorphous materials of the same chemical composition illustrates, more than anything else, why structure must be studied before properties:
| Property | Crystalline Material | Amorphous Material |
|---|---|---|
| Atomic arrangement | Long-range periodic order | Short-range order only, no periodicity |
| Melting behaviour | Sharp, well-defined melting point | Gradual softening over a range of temperatures |
| Electronic properties | Well-defined band structure, band gaps | Localised electronic states, no clear bands |
| Mechanical properties | Anisotropic, slip systems defined | Isotropic, no preferred fracture plane |
| Examples | Silicon, iron, aluminium, quartz | Glass, metallic glass, amorphous silicon |
| Optical properties | Birefringence, defined refractive indices | Isotropic optical behaviour |
The same chemical element — for example, silicon — behaves completely differently in its crystalline form (used in every transistor on the planet) compared to amorphous silicon (used in cheaper thin-film solar cells with lower efficiency). Structure governs behaviour. This is the central lesson of this lecture.
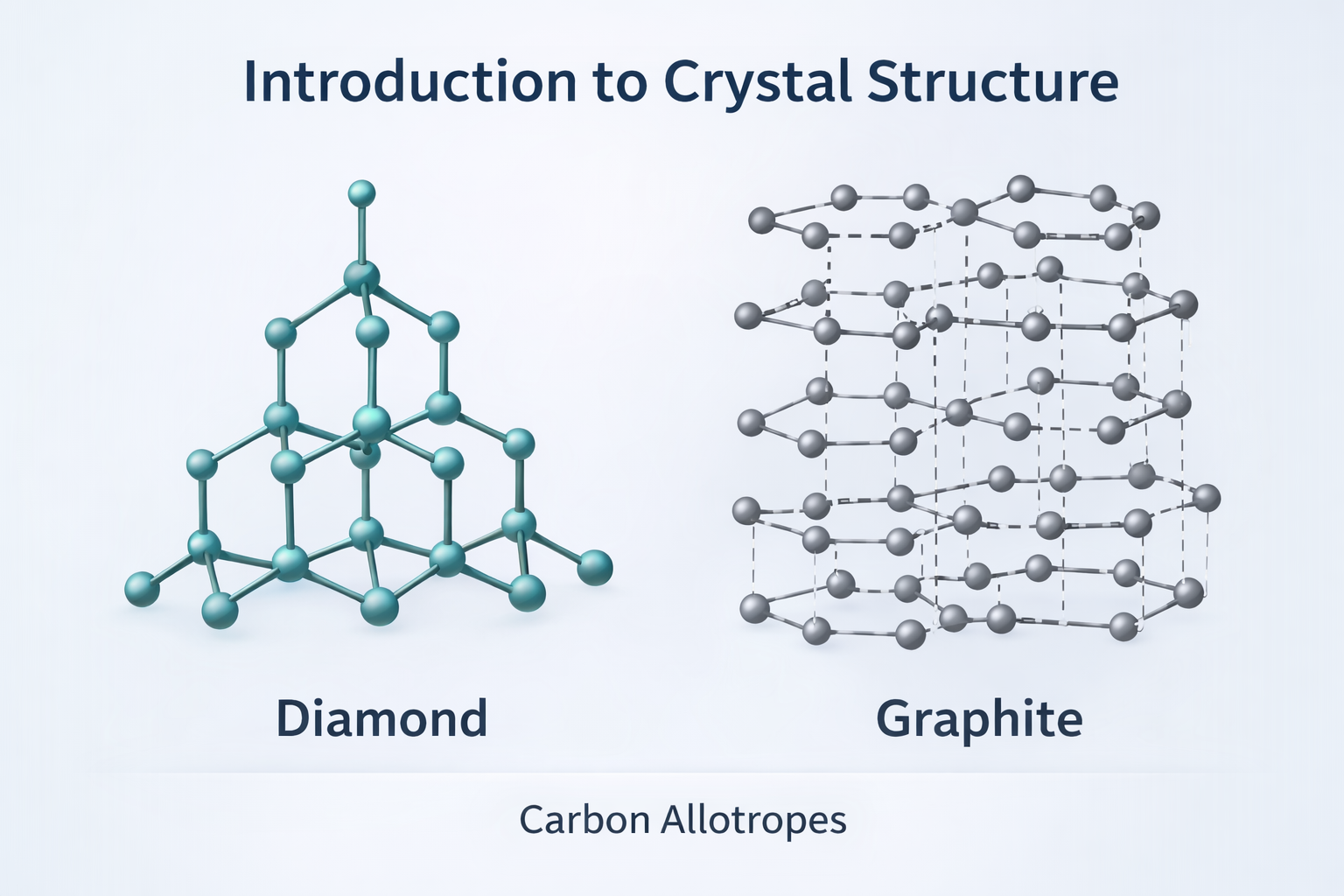
3. A Classical Example: The Carbon Story
The most powerful single demonstration of the structure-property relationship is carbon. Carbon offers us two of the most striking extremes in all of materials science — and both are made from exactly the same atoms.
3.1 Diamond: The Hardest Natural Material
In diamond, each carbon atom forms four strong covalent bonds arranged in a perfect tetrahedron pointing toward its four nearest carbon neighbours. This arrangement repeats identically throughout the entire crystal in three dimensions — the diamond cubic structure. The result of this rigid, three-dimensional network of strong covalent bonds is extraordinary:
- Hardness: Diamond is the hardest natural substance known (Mohs hardness = 10), because no crystal plane slides easily against another — every direction is locked by equally strong bonds
- Thermal conductivity: Diamond conducts heat better than any other natural material (2,000 W/m·K), because the rigid lattice transmits vibrations (phonons) with minimal scattering
- Electrical insulation: Diamond is an electrical insulator — the band gap is 5.47 eV, far too large for electrons to jump across at room temperature
3.2 Graphite: Slippery and Electrically Conducting
In graphite, carbon atoms arrange themselves in flat hexagonal sheets. Within each sheet, three strong covalent bonds per atom give excellent in-plane strength. However, the sheets are held together only by weak van der Waals forces — the same fleeting electrostatic forces that allow geckos to walk on ceilings. The result:
- Softness: Sheets slide easily over one another — which is why graphite is used as a lubricant and in pencils
- Electrical conductivity: The fourth valence electron from each carbon atom is delocalised across the sheets, allowing free electron movement — graphite conducts electricity along the planes
- Anisotropy: Properties differ dramatically parallel and perpendicular to the sheets
The chemical composition in both diamond and graphite is identical: 100% carbon. The atomic mass is identical. The number of electrons per atom is identical. Yet one material is the hardest substance on Earth; the other leaves marks on paper. The difference is entirely and solely due to atomic arrangement.
This example alone justifies why crystal structure must be studied before any advanced materials analysis. There is no shortcut. You cannot predict material behaviour from chemical composition alone.
3.3 Carbon in the 21st Century — Graphene and Beyond
Carbon's structural story continues into the 21st century. Graphene — a single atomic layer of graphite — was isolated in 2004 by Andre Geim and Konstantin Novoselov at the University of Manchester, earning them the Nobel Prize in Physics in 2010. A single atom thick crystal of carbon exhibits: extraordinary strength (200× stronger than steel), perfect electrical conductivity at room temperature, and optical transparency of 97.7%. All of this from one atomic layer of the same hexagonal arrangement as graphite. Structure — at the atomic scale — defines everything.
4. Periodicity and Electronic Behaviour — The Band Theory Connection
When atoms are arranged periodically in a crystal, electrons moving through the material experience a periodic potential energy field — the electric attraction from each positively charged nucleus repeating at regular intervals throughout the lattice. This may sound like a minor detail, but it has consequences so profound that it forms the entire basis of modern electronics.
4.1 Energy Bands and Band Gaps
According to quantum mechanics, when electrons move through a periodic potential, their allowed energies are not continuous — they form allowed energy bands separated by forbidden band gaps. This result, first developed rigorously in the 1920s and 1930s through the work of Felix Bloch, is known as band theory.
- Metals (e.g., copper, iron): Partially filled energy bands — electrons can move freely → electrical conductor. This arises because the band structure allows electrons near the Fermi level to gain energy from an applied field.
- Semiconductors (e.g., silicon, germanium): A moderate band gap (~1 eV) — at room temperature, thermal energy promotes some electrons across the gap → controlled conductivity that can be engineered by doping. Silicon's band gap of 1.12 eV makes it ideal for transistors operating at room temperature.
- Insulators (e.g., diamond, alumina): A large band gap (>3 eV) — electrons cannot cross the gap under normal conditions → electrical insulator.
Here is the critical point: without crystal periodicity, the concept of energy bands would not exist. Band theory is a direct mathematical consequence of the periodic potential created by the crystal lattice. In an amorphous material, this periodicity is absent, and electrons experience a random, disordered potential — which is why amorphous silicon has far lower carrier mobility than single-crystal silicon.
Your mobile phone, laptop processor, solar panel, and LED display all operate because of this quantum mechanical consequence of atomic periodicity. Crystal structure is not an academic abstraction — it is the physical foundation of the digital world.
5. Mechanical Properties and Crystal Geometry
Mechanical behaviour also originates directly from the geometry of atomic arrangement. To understand this, we need to briefly introduce the concept of plastic deformation in crystalline metals.
5.1 Dislocations and Slip Systems
When a metal is bent or stretched permanently (plastically deformed), it does not simply break. Instead, layers of atoms slide over one another along specific crystallographic planes and directions — a process called slip. The sliding occurs along the most densely packed planes in the crystal, because these planes have the greatest spacing between them and the lowest resistance to sliding.
The combination of a specific slip plane and a specific slip direction within that plane is called a slip system. The number of available slip systems determines how easily a metal can deform plastically — which translates directly to its ductility.
- Face-Centred Cubic (FCC) metals (aluminium, copper, gold): 12 slip systems → highly ductile. FCC metals can be drawn into thin wires and rolled into thin sheets without fracturing.
- Body-Centred Cubic (BCC) metals (iron, chromium, tungsten): Up to 48 slip systems geometrically, but slip planes are less densely packed → moderate ductility, higher strength, characteristic ductile-to-brittle transition at low temperatures.
- Hexagonal Close-Packed (HCP) metals (titanium, magnesium, zinc): Only 3 primary basal slip systems → limited ductility, harder to deform, more prone to fracture.
This direct connection between crystal structure type and mechanical behaviour is why materials engineers choose metals for specific applications based on their crystal structure — not just their chemical composition. The structural geometry is a mechanical design parameter.
6. Functional Materials and the Role of Symmetry
In advanced materials science, particularly in the study of functional materials — materials that convert one form of energy into another — crystal symmetry plays a decisive, predictive role. This is where crystallography becomes genuinely powerful as a tool for materials design.
6.1 Symmetry as a Physical Constraint
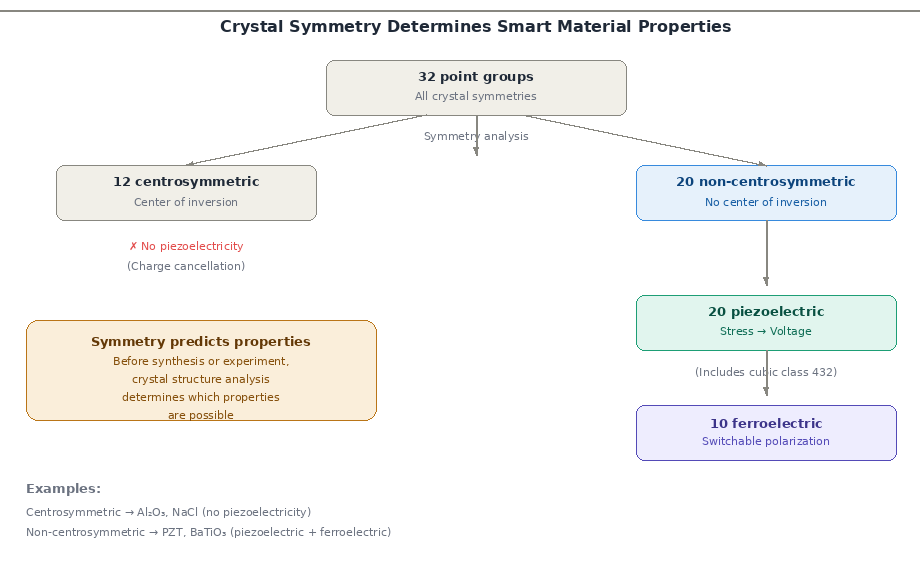
The symmetry of a crystal is not merely a geometric property. It constrains which physical phenomena can occur in that material. This is embodied in a principle known as Neumann's Principle: the physical properties of a crystal must be at least as symmetric as the crystal's point group symmetry.
A practical consequence of this principle:
- Piezoelectricity (conversion of mechanical stress into electric charge) is only possible in crystals that lack a centre of inversion symmetry. Of the 32 crystal point groups, only 20 are non-centrosymmetric and can therefore exhibit piezoelectricity.
- Ferroelectricity (spontaneous, reversible electric polarisation) requires a polar crystal structure — only 10 of the 32 point groups are polar. Materials like barium titanate (BaTiO₃) and lead zirconate titanate (PZT) derive their remarkable piezoelectric and ferroelectric properties directly from their non-centrosymmetric crystal structure.
- Ferromagnetism and multiferroic behaviour depend critically on magnetic space group symmetry.
These conclusions are not empirical guesses — they arise from a rigorous mathematical analysis of crystallographic symmetry. By examining a crystal's space group alone, one can predict whether piezoelectricity, ferroelectricity, or optical activity is even theoretically possible in that material.
This predictive power of crystallographic analysis is precisely what makes it indispensable in materials research. Rather than testing every material empirically, symmetry analysis allows researchers to screen candidates theoretically before synthesising a single sample.
7. Crystal Structure and Modern Technology
Let us now connect everything we have discussed to the concrete technologies that surround you. In every case below, crystal structure is not a background detail — it is the primary engineering variable.
7.1 Semiconductor Technology
The entire semiconductor industry rests on the diamond cubic crystal structure of silicon. The periodic silicon lattice enables band engineering, controlled doping with atoms like phosphorus or boron, and the precise control of charge carrier mobility that makes transistors possible. A single modern silicon chip contains over 100 billion transistors — each one functional because of the crystal structure of the substrate on which it was built. Without crystalline silicon, the digital age would not exist.
7.2 Lithium-Ion Batteries
In lithium-ion batteries — which power your phone, laptop, and electric vehicle — energy storage depends on the ability of lithium ions to move rapidly through specific crystallographic channels in the cathode material (typically layered lithium cobalt oxide, LiCoO₂, or olivine lithium iron phosphate, LiFePO₄). The diffusion pathways are defined by the crystal structure. Optimising battery energy density and charging speed is fundamentally an exercise in crystal structure engineering.
7.3 Photovoltaic Materials
In solar cells, the efficiency with which light is absorbed and converted to electricity depends on the band gap of the absorber material — which is determined by its crystal structure. The perovskite crystal structure (ABX₃ formula type) has emerged as one of the most promising solar cell materials because its band gap can be tuned by changing the chemical composition while keeping the crystal structure type constant.
7.4 Magnetic Data Storage
The read/write heads and magnetic storage layers in hard drives exploit the magnetocrystalline anisotropy of crystalline magnetic materials — the property that magnetisation prefers certain crystallographic directions over others. Without this structural anisotropy, stable magnetic data storage would be impossible.
8. 2025 Frontiers: Crystal Symmetry in Quantum Materials
The relationship between crystal structure and material properties has taken on an entirely new dimension in the past decade with the discovery and study of topological quantum materials. This is one of the most active frontiers in condensed matter physics and materials science, and it demonstrates that our understanding of crystal symmetry continues to open new doors.
8.1 Topological Insulators — Where Crystal Symmetry Meets Quantum Mechanics
A topological insulator is a material that behaves as an electrical insulator in its bulk interior but conducts electricity perfectly along its surface or edges — and this surface conductivity is protected by crystal symmetry. Damage the surface, introduce disorder, scatter electrons — the conducting surface states survive because the underlying crystal symmetry guarantees their existence. This robustness is qualitatively different from conventional conductors.
The conducting surface states in topological insulators arise from a property of the electronic band structure called band topology — a mathematical property that describes how electron wavefunctions twist and connect as they traverse the Brillouin zone (the reciprocal space representation of the crystal lattice). The band topology is directly determined by the crystal symmetry.
In a landmark result published in Nature Communications in 2025, researchers from the University of Jyväskylä and Aalto University experimentally realised a two-dimensional topological crystalline insulator in bilayer tin telluride (SnTe) grown on a NbSe₂ substrate. This material had been theoretically predicted for over a decade but remained experimentally unrealised due to materials challenges.
The conducting edge states, protected by the crystal lattice symmetry of the SnTe film, formed within a large band gap exceeding 0.2 eV — making them potentially robust at room temperature. Furthermore, the topological properties could be tuned by applying strain to the crystal lattice — demonstrating that controlling crystal structure gives direct control over quantum electronic properties.
This discovery exemplifies the deepest level at which crystal structure governs material behaviour: not just conventional electrical or mechanical properties, but the quantum topology of electronic states itself.
8.2 AI-Accelerated Crystal Structure Discovery
In another major development published in npj Quantum Materials in 2025, researchers used deep generative machine learning models to discover 20 new topological materials — including topological insulators and semimetals — by computationally generating novel crystal structures and screening them for topological properties. This approach, which uses crystal symmetry analysis as the primary screening criterion, marks a new era in which artificial intelligence, guided by crystallographic principles, dramatically accelerates materials discovery.
These examples from the cutting edge of research confirm what we stated at the beginning: crystal structure is not a topic to be studied in the first week and then set aside. It is the foundation that keeps generating new physics and new technologies across the entire history of materials science.
9. How We Study Crystal Structures — An Introduction to XRD
Given that crystal structure is so important, how do scientists actually determine it? The primary tool is X-ray diffraction (XRD), a technique discovered by Max von Laue in 1912 — a discovery so significant that it earned the Nobel Prize in Physics in 1914.
9.1 The Principle of X-ray Diffraction
When X-rays — electromagnetic radiation with wavelengths comparable to atomic spacings (0.1–0.2 nm) — are directed at a crystal, they interact with the periodic array of atoms and scatter in specific directions. Because the atoms are arranged periodically, the scattered X-ray waves interfere constructively (add together) only in specific directions determined by the lattice spacing and the X-ray wavelength. This phenomenon is described by Bragg's Law:
nλ = 2d sin θ
Where n is an integer (diffraction order), λ is the X-ray wavelength, d is the spacing between atomic planes, and θ is the angle of incidence. By measuring the angles at which constructive interference occurs, scientists extract the lattice parameters, atomic positions, and complete symmetry of the crystal structure.
9.2 What XRD Reveals
From XRD patterns, materials scientists can determine:
- The unit cell dimensions — the basic repeating block of the crystal
- The positions of every atom within the unit cell
- The crystal symmetry — which space group the structure belongs to
- The phase composition — which crystal phases are present in a mixture
- The crystallite size from peak broadening (Scherrer equation)
Without XRD, modern materials engineering would lack its primary structural characterisation tool. Every crystal structure we discuss in this tutorial series has been determined — or verified — using X-ray diffraction.
10. What This Lecture Series Covers Next
Now that you understand why crystal structure matters, the rest of this series will systematically build the mathematical and physical framework needed to describe, analyse, and predict crystal structures rigorously. Here is what each lecture covers:
At a deeper level, a crystal is more than a geometric arrangement of atoms. It is a periodic energy landscape that shapes electron motion, lattice vibrations (phonons), thermal transport, defect behaviour, and — as the 2025 discoveries remind us — quantum topological properties. The periodic boundary conditions imposed by crystal symmetry constrain physical laws within the material. Crystal structure does not merely influence properties: it defines the framework within which all physical processes inside a material occur.
11. Practice Questions
- (a) Diamond contains more carbon atoms per unit volume
- (b) The atomic arrangement (crystal structure) differs between diamond and graphite ✔
- (c) The isotopes of carbon differ between the two materials
- (d) Diamond is crystalline and graphite is amorphous
- (a) The chemical bonding type (ionic, covalent, metallic)
- (b) The periodic arrangement of atoms creating a repeating potential field ✔
- (c) The density of the material
- (d) The temperature at which the crystal was formed
- (a) High atomic packing factor
- (b) Large unit cell volume
- (c) Absence of a centre of inversion symmetry (non-centrosymmetric structure) ✔
- (d) High melting point of the material
- (a) The high purity of the crystal — absence of impurities
- (b) The large thickness of the surface layer
- (c) The symmetry of the crystal lattice and the non-trivial band topology ✔
- (d) The elevated temperature at which the material is operated
- (a) Scanning electron microscopy (SEM)
- (b) Atomic force microscopy (AFM)
- (c) X-ray diffraction (XRD) ✔
- (d) Differential scanning calorimetry (DSC)
- (a) Carbon can only exist in amorphous forms at the nanoscale
- (b) Even a single atomic layer retains a well-defined crystal structure with extraordinary properties ✔
- (c) Crystal structure is irrelevant below 5 atomic layers of thickness
- (d) Van der Waals forces are stronger than covalent bonds
12. Key Takeaways
- THE CORE PRINCIPLE: The properties of a material are determined not only by its chemical composition, but primarily by how its atoms are arranged in space — the crystal structure.
- DEFINITION OF A CRYSTAL: A crystalline solid has atoms arranged in a periodic, repeating pattern extending in three dimensions. This periodicity creates symmetry, which governs physical responses.
- CARBON DEMONSTRATION: Diamond (3D tetrahedral network) is the hardest material and an insulator. Graphite (layered hexagonal sheets) is soft and electrically conducting. Same atoms, completely different structures, completely different properties.
- ELECTRONIC BEHAVIOUR: Crystal periodicity creates a repeating potential field that produces electronic energy bands and band gaps — the physical basis of conductors, semiconductors, and insulators.
- MECHANICAL BEHAVIOUR: The number and geometry of slip systems — determined by crystal structure type (FCC, BCC, HCP) — directly controls ductility, strength, and fracture resistance.
- SYMMETRY AND FUNCTION: Crystal symmetry determines which physical phenomena are possible. Only non-centrosymmetric crystals can be piezoelectric; only polar structures can be ferroelectric.
- MODERN TECHNOLOGY: Semiconductors, batteries, solar cells, magnetic storage, and quantum devices all depend on crystal structure as a primary design parameter.
- QUANTUM FRONTIER (2025): Topological insulators, whose conducting surface states are protected by crystal symmetry, represent a new paradigm in materials science enabled by crystallographic understanding.
- EXPERIMENTAL TOOL: X-ray diffraction (XRD), described by Bragg's Law (nλ = 2d sinθ), is the primary technique for determining crystal structures experimentally.
- WHAT COMES NEXT: The next lecture defines lattices, bases, and unit cells — the mathematical building blocks for describing all crystal structures rigorously.
References
All references are in IEEE citation style. All sources are peer-reviewed journals, internationally recognised textbooks, or authoritative academic institutions.
- C. Kittel, Introduction to Solid State Physics, 8th ed. Hoboken, NJ, USA: John Wiley & Sons, 2005, ch. 1–2. [Wiley] — Primary reference for crystal periodicity, band theory, and Bloch's theorem.
- W. D. Callister Jr. and D. G. Rethwisch, Materials Science and Engineering: An Introduction, 10th ed. Hoboken, NJ, USA: John Wiley & Sons, 2018, ch. 3. [Wiley] — Standard undergraduate reference for crystal structure fundamentals, crystal systems, and Bravais lattices.
- N. W. Ashcroft and N. D. Mermin, Solid State Physics. Philadelphia, PA, USA: Harcourt College Publishers, 1976, ch. 4–8. — Classic graduate-level reference for band theory, Brillouin zones, and electronic structure arising from crystal periodicity.
- B. D. Cullity and S. R. Stock, Elements of X-Ray Diffraction, 3rd ed. Upper Saddle River, NJ, USA: Pearson Prentice Hall, 2001. — Definitive reference for X-ray diffraction principles, Bragg's Law derivation, and structure determination methods.
- International Union of Crystallography (IUCr), "Teaching Pamphlets: Crystal Symmetry and Space Groups," IUCr Education Pamphlets, no. 9. Chester, UK: IUCr, 2002. [iucr.org — full text] — Authoritative reference for the 7 crystal systems, 14 Bravais lattices, and 230 space groups.
- K. S. Novoselov et al., "Electric field effect in atomically thin carbon films," Science, vol. 306, no. 5696, pp. 666–669, Oct. 2004, doi: 10.1126/science.1102896. [DOI: 10.1126/science.1102896] — Nobel Prize-winning paper reporting the isolation of graphene — the single-layer carbon crystal demonstrating extreme structure-property relationships.
- L. Jing et al., "Strain-induced two-dimensional topological crystalline insulator in bilayer SnTe," Nature Communications, vol. 16, Art. no. 6304, Jul. 2025, doi: 10.1038/s41467-025-67520-y. [Nature Communications — Open Access] — 2025 experimental realisation of a 2D topological crystalline insulator with edge states protected by crystal lattice symmetry.
- Y. Luo, C. Liu, and S. Ji, "Discovery of new topological insulators and semimetals using deep generative models," npj Quantum Materials, vol. 10, Art. no. 22, Jan. 2025, doi: 10.1038/s41535-025-00731-0. [npj Quantum Materials — Open Access] — AI-guided crystal structure generation used to discover 20 new topological quantum materials.
- R. Verma and S. K. Rout, "Frequency-dependent ferro–antiferro phase transition and internal bias field influenced piezoelectric response of donor and acceptor doped bismuth sodium titanate ceramics," J. Appl. Phys., vol. 126, no. 9, Art. no. 094103, Sep. 2019, doi: 10.1063/1.5111505. [AIP — DOI: 10.1063/1.5111505] — Author's peer-reviewed research demonstrating how crystal structure and symmetry determine ferroelectric and piezoelectric behaviour in perovskite ceramics.
- R. Verma and S. K. Rout, "The Mystery of Dimensional Effects in Ferroelectricity," in Recent Advances in Multifunctional Perovskite Materials, P. Sharma and A. Kumar, Eds. London, UK: IntechOpen, 2022, doi: 10.5772/intechopen.104435. [IntechOpen — DOI: 10.5772/intechopen.104435] — Author's book chapter on crystal structure effects in functional perovskite materials relevant to sections 6 and 7.
This is Lecture 01 of the Crystal Structure Hub Series at AdvanceMaterialsLab.com
Next Lecture: Lattice and Basis in Crystallography →
© 2026 AdvanceMaterialsLab.com — Dr. Rolly Verma | All rights reserved
Dr. Rolly Verma
Explore More from Advance Materials Lab
Continue expanding your understanding of material behavior through our specialized resources on ferroelectrics and phase transitions. Each guide is designed for research scholars and advanced learners seeking clarity, precision, and depth in the field of materials science and nanotechnology.
🔗 Recommended Reads and Resources:
Ferroelectrics Tutorials and Research Guides — Comprehensive tutorials covering polarization, hysteresis, and ferroelectric device characterization.
Workshops on Ferroelectrics (2025–2027) — Upcoming training sessions and research-oriented workshops for hands-on learning.
Glossary — Ferroelectrics and Phase Transitions — Concise explanations of key terminologies to support your study and research work.
If you notice any inaccuracies or have constructive suggestions to improve the content, I warmly welcome your feedback. It helps maintain the quality and clarity of this educational resource. You can reach me at: advancematerialslab27@gmail.com
