Crystal Structure
Raman Spectroscopy
Research Careers
Ferroelectrics & Testing Systems
Miller Indices: How to Find (hkl) Planes, Bar Notation & Miller-Bravais — Complete Guide
The Universal Language for Naming Planes and Directions in Crystals — Step-by-Step with Worked Examples By Dr. Rolly Verma | AdvanceMaterialsLab.com | March 2026 | B.Sc. / M.Sc. Materials Science & EngineeringSeries: Crystal Structure Hub | Level: Undergraduate / Postgraduate | Prerequisites: Crystal Structure Intro, Unit Cell & Lattice Parameters
Reading time: 40 minutes | Includes: 3-step method, negative indices (bar notation), families of planes, crystallographic directions, Miller-Bravais for hexagonal, d-spacing formulas, 7 worked examples, 8 MCQs, key takeaways
SEO Keywords: Miller indices how to find, Miller indices of a plane steps, Miller-Bravais indices hexagonal, negative Miller indices bar notation, Miller indices worked examples, crystallographic planes directions, hkl notation crystal
1. Why Miller Indices? The Problem They Solve
Throughout the Crystal Structure Hub series, we have encountered expressions like "atoms touch along the face diagonal ⟨110⟩", "slip occurs on the {111} planes", "BCC atoms touch along [111]", and "XRD peaks are labelled by (hkl) indices". In every tutorial — from the BCC structure to the FCC structure — these symbols appeared without a dedicated explanation. This lecture provides that explanation.
The challenge in crystallography is this: a crystal contains an infinite number of planes (and directions) passing through lattice points, oriented in every possible direction. To communicate unambiguously about a specific plane or direction — across all languages, all countries, all textbooks — we need a universal, compact notation system. That system is Miller indices, developed by the British mineralogist William Hallowes Miller in 1839.
- A compact, unambiguous 3-integer notation (h, k, l) that uniquely identifies any crystallographic plane in a cubic, tetragonal, or orthorhombic crystal.
- A consistent way to name crystallographic directions [u, v, w] and families of equivalent planes {h, k, l}.
- The mathematical bridge between crystal geometry and XRD diffraction patterns — every peak in an XRD pattern is labelled by its (hkl) Miller indices.
- The language for describing slip systems, cleavage planes, grain boundaries, thin film orientations, and electronic surface states in semiconductor devices.
2. The Core Concept — Intercepts and Reciprocals
Before we learn the steps, we need to understand the underlying idea. A crystallographic plane is identified by how it intersects the three crystal axes (a, b, c) of the unit cell. Specifically, we record at what multiple of the lattice parameter each axis is cut — these are the intercepts.
However, we use the reciprocals of the intercepts rather than the intercepts themselves. This choice is deliberate and elegant — it solves two problems at once:
Reason 1 — Handling parallel planes: A plane that is parallel to an axis never intersects it — its intercept would be infinity (∞). The reciprocal of infinity is 0, which is a clean, manageable integer. Without reciprocals, we could not write a notation for parallel planes.
Reason 2 — Connection to diffraction: The reciprocals of intercepts are directly proportional to the components of the normal vector to the plane. This connects Miller indices naturally to the reciprocal lattice — the mathematical structure that underlies X-ray diffraction theory. Every XRD peak occurs at a point in reciprocal space labelled (hkl).
3. How to Find Miller Indices of a Plane — The 3-Step Procedure
This is the most important section for students asking "Miller indices how to find" and "Miller indices of a plane steps". Once you master this 3-step procedure, you can find the Miller indices of any plane in any crystal system.
Express each intercept as a multiple of the corresponding lattice parameter. If the plane is parallel to an axis, write ∞ for that intercept. If the plane passes through the origin, shift the origin to a nearby equivalent lattice point before reading intercepts.
Example: a plane cuts the x-axis at 1, the y-axis at 2, and is parallel to the z-axis → intercepts are (1, 2, ∞)
Invert each intercept value. The reciprocal of ∞ is 0. The reciprocal of 1 is 1. The reciprocal of 2 is 1/2. Do not simplify or round yet.
Example: reciprocals of (1, 2, ∞) → (1/1, 1/2, 1/∞) = (1, 1/2, 0)
Find the lowest common denominator of all fractions, multiply through, and confirm all results are integers. Write the final indices in parentheses: (hkl).
Example: multiply (1, 1/2, 0) by 2 → (2, 1, 0) → Miller indices are (210)
Miller indices are always computed with respect to a specific origin. If the plane passes through the chosen origin, you must shift the origin to any other equivalent lattice point (typically move it to an adjacent unit cell corner) before applying the 3 steps. The resulting indices will be the same regardless of which equivalent origin you choose — by the definition of a lattice, all lattice points have identical environments.
4. Negative Miller Indices — Bar Notation
One of the most commonly searched topics: "negative Miller indices bar notation". When the reciprocal of an intercept is a negative number — which occurs when the plane intercepts an axis on the negative side of the origin — the corresponding Miller index is negative. In standard crystallographic notation, a negative index is written with a bar (overline) above the number, not with a minus sign.
A negative Miller index is written with a bar above the digit:
−1 is written as 1 (read: "bar one")
−2 is written as 2 (read: "bar two")
So a plane with indices (1, −1, 0) is written as (110) and pronounced "one bar-one zero". In typed text (HTML, papers), this is often written as (1-10) or (1$\bar{1}$0) in LaTeX.
4.1 When Do Negative Indices Appear?
A negative index arises when a plane intercepts an axis at a negative coordinate — i.e., on the opposite side of the origin from the positive axis direction. In practice this occurs when you are looking at planes in the lower, rear, or left portions of the unit cell.
Example: A plane intercepts the x-axis at −1, y-axis at 1, z-axis at 1.
Intercepts: −1, 1, 1
Reciprocals: −1, 1, 1
Integers: −1, 1, 1
Miller indices: (1̄11) ← read "bar-one, one, one"
The planes (111) and (1̄11) are not the same plane. They are parallel planes on opposite sides of the origin, related by inversion. Together they form a pair of parallel planes bounding the unit cell in that direction. However, (111) and (1̄1̄1̄) are the same plane — just described from opposite sides — and represent the same physical crystallographic plane.
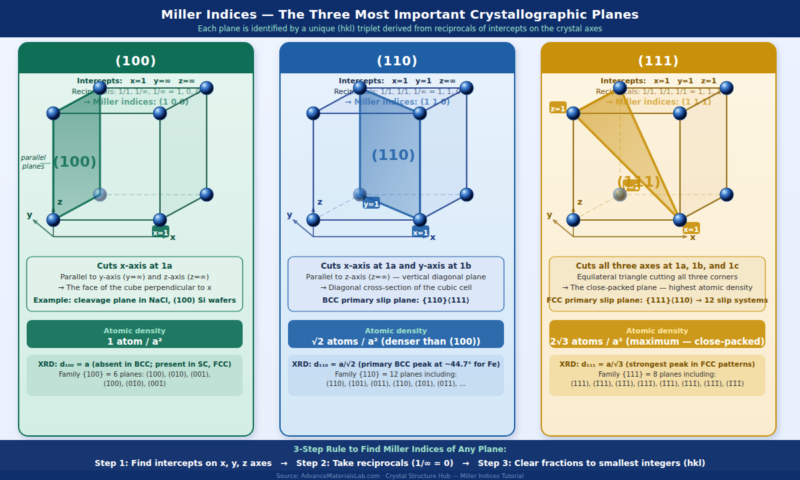
5. Special Planes — The Three Most Important in Cubic Crystals
The three most fundamental planes in the cubic crystal system appear repeatedly throughout materials science. Every student must be able to identify, draw, and derive these immediately.
| Plane | Intercepts (a, b, c) | Reciprocals | Miller Indices | Description & Significance |
|---|---|---|---|---|
| (100) | 1, ∞, ∞ | 1, 0, 0 | (100) | The face of the cube perpendicular to the x-axis. Contains b and c axes. Cleavage plane in many ionic crystals (NaCl). |
| (110) | 1, 1, ∞ | 1, 1, 0 | (110) | Diagonal cross-section of the cube, containing the c-axis. BCC atoms touch along the face diagonal of this plane. BCC primary slip plane. |
| (111) | 1, 1, 1 | 1, 1, 1 | (111) | The close-packed plane — cuts all three axes equally, forming an equilateral triangle cross-section. FCC and HCP slip plane. Highest atomic density in FCC. |
| (200) | ½, ∞, ∞ | 2, 0, 0 | (200) | Parallel to (100), halfway through the unit cell. Appears as a separate peak in XRD patterns of FCC metals. |
| (210) | 1, 2, ∞ | 1, ½, 0 | (210) | Cuts the x-axis at 1 and y-axis at 2, parallel to z. Lower-symmetry plane, less dense. |
In the FCC crystal structure, the {111} planes are the most densely packed planes — atoms in these planes are arranged in a perfect hexagonal close-packed pattern. This is why FCC slip occurs on {111} planes (4 independent orientations), giving FCC metals their 12 slip systems and exceptional ductility. In HCP metals, the basal plane (0001) is equivalent to the (111) plane in FCC — both are hexagonally close-packed layers. Understanding the (111) plane is understanding the origin of ductility in metals.
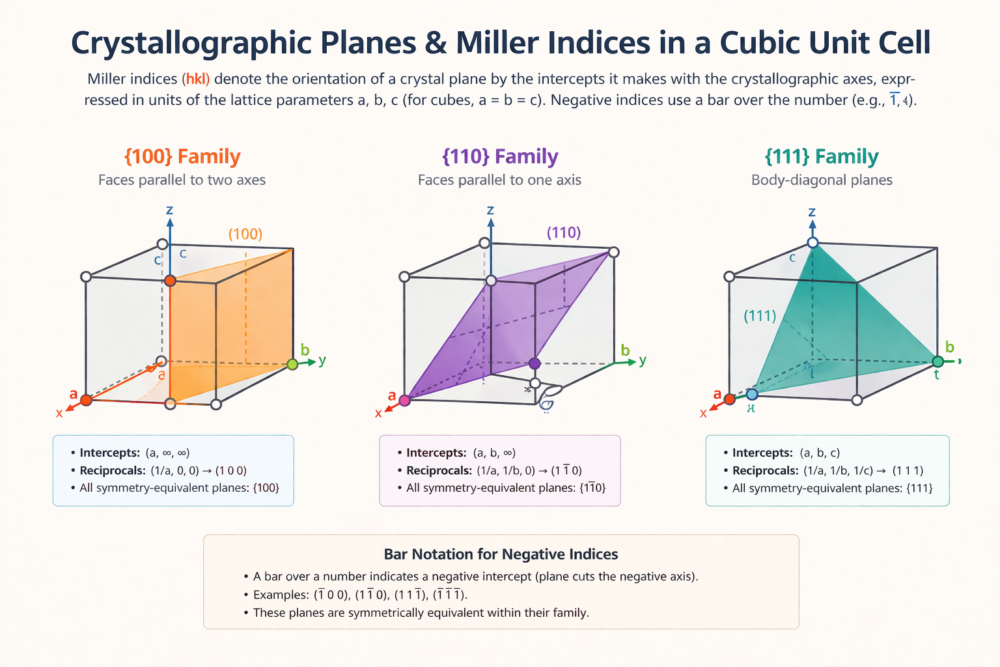
6. Families of Planes {hkl} and Crystallographic Equivalence
In a crystal with cubic symmetry, many planes that appear geometrically different are actually crystallographically equivalent — they have identical atomic density, identical spacing, and identical physical properties. The symmetry of the crystal maps one onto another through rotation or reflection operations. Such a group of equivalent planes is called a family of planes and is denoted by the curly bracket notation {hkl}.
6.1 The {100} Family — Six Equivalent Cube Faces
Family {100} contains 6 equivalent planes:
(100), (010), (001), (1̄00), (01̄0), (001̄)
In a cubic crystal, these six planes are the six faces of the cube. By the cubic symmetry, every face has the same atomic arrangement, the same d-spacing, and the same physical properties. There is no distinction between "the top face" and "the front face" in a perfect cubic crystal — they are crystallographically identical.
6.2 The {110} Family — Twelve Diagonal Planes
Family {110} contains 12 equivalent planes:
(110), (101), (011), (1̄10), (1̄01), (01̄1),
(11̄0), (101̄), (011̄), (1̄1̄0), (1̄01̄), (01̄1̄)
6.3 The {111} Family — Eight Close-Packed Planes
Family {111} contains 8 equivalent planes:
(111), (11̄1̄), (1̄11̄), (1̄1̄1), (1̄1̄1̄), (1̄11), (11̄1), (111̄)
- (hkl) — a specific plane, e.g. (111) is one particular close-packed plane
- {hkl} — a family of all crystallographically equivalent planes, e.g. {111} includes all 8 close-packed planes in cubic
- [uvw] — a specific direction, e.g. [110] is one face diagonal direction
- ⟨uvw⟩ — a family of all equivalent directions, e.g. ⟨110⟩ includes all 12 face diagonal directions in cubic
7. Crystallographic Directions [uvw] and Families ⟨uvw⟩
Miller indices describe planes. Crystallographic direction indices describe directions — vectors connecting two lattice points. The procedure for finding direction indices is different from finding plane indices.
7.1 How to Find Direction Indices [uvw]
Choose the nearest lattice point in the desired direction. If the vector does not start at the origin, translate it (parallel to itself) until it does.
Express the endpoint coordinates (x, y, z) as multiples of the lattice parameters. These are already the direction indices — there is no reciprocal step for directions.
If the result contains fractions, multiply through by the smallest integer that removes all fractions. Negative components are written with a bar.
7.2 Important Directions in the Cubic System
| Direction | Vector from Origin to Point | Description | Significance |
|---|---|---|---|
| [100] | (1,0,0) | Along the x (a) axis | Cube edge direction. SC touching direction. |
| [110] | (1,1,0) | Face diagonal in x-y plane | FCC touching direction. FCC/BCC slip direction. |
| [111] | (1,1,1) | Body diagonal | BCC touching direction. BCC slip direction. |
| [1̄10] | (−1,1,0) | Face diagonal, negative x | Equivalent to [110] in cubic — same family ⟨110⟩ |
| [210] | (2,1,0) | Runs 2 units along x, 1 along y | Step edge direction in surface science |
In the cubic crystal system only, the direction [hkl] is always perpendicular (normal) to the plane (hkl) of the same indices. This is a special property of cubic symmetry and is not true in tetragonal, hexagonal, or other lower-symmetry systems. For example, the direction [111] is perpendicular to the plane (111), and [110] is perpendicular to (110). This makes visualisation and calculation much simpler in cubic crystals.
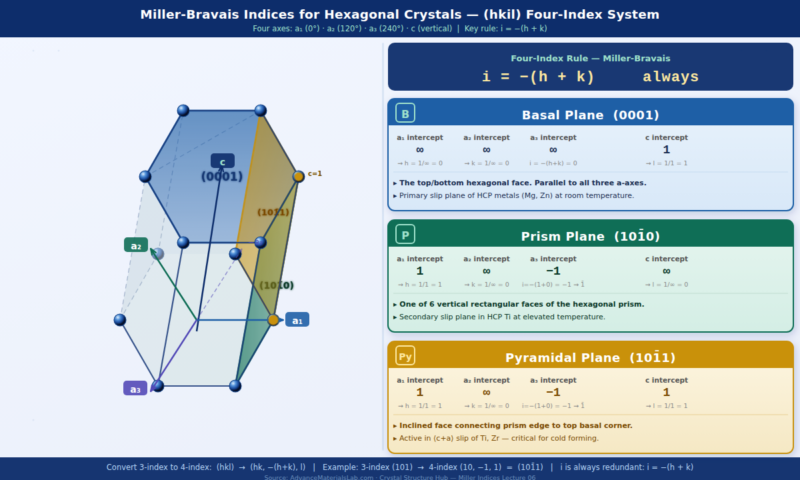
8. Miller-Bravais Indices for Hexagonal Crystals (hkil)
Hexagonal crystals — which includes the HCP structure adopted by Mg, Ti, Zn, Co, and Zr — present a special challenge. The standard 3-index Miller notation (hkl) technically works for hexagonal crystals, but it obscures the 6-fold symmetry of the hexagonal lattice. Equivalent planes that should have the same physical properties end up with apparently different indices. To solve this, the Miller-Bravais 4-index notation (hkil) is used for hexagonal crystals.
8.1 The Four-Axis System for Hexagonal Crystals
In the Miller-Bravais system, the hexagonal unit cell is described using four axes rather than three:
- a₁, a₂, a₃ — three axes in the basal plane, separated by 120° from each other
- c — the vertical axis, perpendicular to the basal plane
The three basal-plane axes are not independent — any two determine the third. The relationship between the four indices is:
i = −(h + k)
The four Miller-Bravais indices are: (hkil) where i = −(h+k)
8.2 Converting from 3-Index to 4-Index Notation
To convert a 3-index notation (hkl) to Miller-Bravais 4-index (hkil), simply insert i = −(h + k) as the third index:
3-index: (hkl) → 4-index: (hk, −(h+k), l)
Examples:
(100) → (10, −1, 0) = (101̄0) ← a prism face of the hexagon
(001) → (00, 0, 1) = (0001) ← the basal plane
(011) → (01, −1, 1) = (011̄1) ← a pyramidal plane
8.3 The Most Important Planes in HCP Crystals
| Plane Name | Miller-Bravais | 3-Index equiv. | Description & Significance |
|---|---|---|---|
| Basal plane | (0001) | (001) | The top/bottom hexagonal face. Primary slip plane in HCP metals. Densest plane. |
| Prism plane (1st order) | (101̄0) | (100) | Vertical rectangular face. Secondary slip in HCP at elevated temperature. |
| Prism plane (2nd order) | (112̄0) | (110) | Wider vertical face. Contains the ⟨1120⟩ close-packed directions. |
| Pyramidal plane (1st order) | (101̄1) | (101) | Inclined face. ⟨c+a⟩ slip at high temperature. Critical for Ti deformation. |
| Pyramidal plane (2nd order) | (112̄2) | (112) | Steeper inclined face. Operative in strongly deformed HCP metals. |
With 3-index notation, the six prism faces of a hexagonal crystal have indices (100), (010), (1̄10), (1̄00), (01̄0), and (110) — they all look different even though they are physically identical. With 4-index Miller-Bravais notation, all six have the form (hkh+k0) with h and k permuted cyclically — the equivalence is immediately visible. This is the entire reason for using 4 indices for hexagonal crystals, as documented by the International Union of Crystallography.
9. Interplanar Spacing d_hkl — The Link to XRD
Once the Miller indices of a plane are known, the interplanar spacing d_hkl (the perpendicular distance between adjacent parallel planes of the same (hkl) family) can be calculated. This quantity is what directly determines the angles at which X-ray diffraction peaks appear through Bragg's Law: nλ = 2d sinθ.
The formula for d_hkl depends on the crystal system. For the three most common systems:
Cubic system (a = b = c, all angles 90°):
d_hkl = a / √(h² + k² + l²)
Tetragonal system (a = b ≠ c, all angles 90°):
1/d²_hkl = (h² + k²)/a² + l²/c²
Hexagonal system (a = b ≠ c, γ = 120°):
1/d²_hkl = (4/3) × (h² + hk + k²)/a² + l²/c²
When you record an XRD pattern of a cubic crystal and observe a peak at angle 2θ, you calculate d = λ/(2 sinθ) from Bragg's Law. Then from d = a/√(h²+k²+l²), you extract the lattice parameter a. The process of assigning (hkl) indices to each peak — called indexing — is the first step in every crystal structure determination by XRD. The complete packing fraction and density calculations covered in our Packing Fraction tutorial all begin with the lattice parameter a determined in exactly this way.
For BCC and FCC structures, not all (hkl) planes produce XRD peaks — some are systematically absent due to destructive interference. These systematic absences follow selection rules, as described in detail by the IUCr guide to powder diffraction:
BCC reflection conditions: (h + k + l) must be even → allowed peaks
FCC reflection conditions: h, k, l must be all-odd or all-even → allowed peaks
BCC allowed: (110), (200), (211), (220), (310), (222)...
FCC allowed: (111), (200), (220), (311), (222), (400)...
10. Why Miller Indices Matter — Engineering Applications
Miller indices are not an abstract academic notation — they encode physically measurable properties that engineers use every day.
10.1 Slip Systems in Plasticity
Every slip system described in our BCC and FCC tutorials is written using Miller indices: {111}⟨110⟩ for FCC, {110}⟨111⟩ for BCC. The specific planes and directions involved determine whether a material is ductile or brittle, how it work-hardens, and how it fractures.
10.2 Semiconductor Surface Orientation
The surface properties of silicon wafers depend critically on their crystallographic orientation. Silicon wafers used in MOSFET transistors are typically (100)-oriented because this surface has the lowest interface state density with SiO₂. Bipolar transistors prefer (111) orientation. The crystal orientation of silicon wafers is specified by their Miller indices and is one of the primary parameters on a wafer specification sheet.
10.3 Thin Film Epitaxy and Texture
When crystalline thin films are grown on substrates, they grow preferentially with a specific crystallographic plane parallel to the substrate surface — called the preferred orientation or texture. This texture is described by Miller indices. For example, copper thin films grown by sputtering typically develop a strong (111) texture — the close-packed plane lies parallel to the substrate surface because this minimises surface energy.
10.4 Cleavage and Fracture
Crystalline materials preferentially fracture along specific crystallographic planes — their cleavage planes — which have low atomic density and weak inter-planar bonding. NaCl cleaves perfectly on {100} planes. Diamond cleaves on {111} planes — the close-packed planes with the lowest surface energy. Mica cleaves on (001) — the basal plane of its layered monoclinic structure. Knowledge of Miller indices allows prediction of fracture behaviour before any mechanical testing.
11. Worked Examples — Step by Step
Step 1 — Intercepts: 1, 2, 3
Step 2 — Reciprocals: 1/1, 1/2, 1/3 = 1, 0.5, 0.333
Step 3 — LCD = 6; multiply all by 6: 6, 3, 2
Miller indices: (632)
Verify: all indices are positive integers — ✔. No common factor other than 1 — ✔. Final answer: (632)
Step 1 — Intercepts: ½, ∞, ∞
Step 2 — Reciprocals: 2, 0, 0
Step 3 — Already integers (no fractions)
Miller indices: (200)
This plane is parallel to both b and c, cutting a at half-lattice-parameter distance. It is the (200) plane — the plane halfway through the cubic cell, parallel to the (100) face.
Step 1 — Intercepts: +1, −1, ∞
Step 2 — Reciprocals: +1, −1, 0
Step 3 — Already integers
Miller indices: (1 1̄ 0) ← "one bar-one zero"
The negative y-intercept gives index −1, written with a bar: 1̄. This is a valid (110)-family plane at an angle in the x-y plane.
The endpoint in units of (a, b, c) is: (1, 1, 1)
No fractions to clear. No common factor.
Direction indices: [111]
The body diagonal direction in a cubic crystal is [111]. This is the touching direction in the BCC structure (4R = a√3), and the slip direction in BCC metals.
Given: h = 1, k = 1, l = 1
i = −(h + k) = −(1 + 1) = −2
Miller-Bravais indices: (hkil) = (11, −2, 1) = (112̄1)
The plane (111) in hexagonal becomes (112̄1) in 4-index notation. This is a first-order pyramidal plane — important for understanding non-basal slip in titanium and zirconium alloys.
Cubic formula: d_hkl = a / √(h² + k² + l²)
For (110): h = 1, k = 1, l = 0
d_110 = 0.2866 / √(1² + 1² + 0²)
= 0.2866 / √2
= 0.2866 / 1.4142
= 0.2027 nm = 2.027 Å
Using Bragg's Law with CuKα (λ = 0.15406 nm):
2θ = 2 × arcsin(λ / 2d) = 2 × arcsin(0.15406 / 0.4054)
= 2 × arcsin(0.3800) = 2 × 22.33° = 44.66° ✔
This is the primary XRD peak of BCC iron at 2θ ≈ 44.7° — one of the most commonly referenced diffraction peaks in steel research.
The plane passes through x = 1 (on the x-axis), y = 1, z = 1.
Intercepts: 1, 1, 1
Reciprocals: 1, 1, 1
Miller indices: (111) ← the close-packed plane in FCC
This is the (111) plane — the most important plane in FCC crystal mechanics and the FCC slip plane. It cuts all three axes at unit distance, forming an equilateral triangle inside the cube.
12. Series Navigation
13. Practice MCQs
- (a) (211)
- (b) (100)
- (c) (100) — wait: reciprocals of (2, ∞, ∞) = (1/2, 0, 0); multiply by 2 → (100). So (100). ✔
- (d) (200) — only if the intercept were ½, not 2
- (a) A larger-than-unit intercept
- (b) A negative index, arising from an intercept on the negative side of the origin ✔
- (c) A fractional index that has not been cleared
- (d) A direction index, not a plane index
- (a) [uvw] lies within the plane (hkl)
- (b) [uvw] is perpendicular (normal) to the plane (hkl) ✔ — true only in cubic systems
- (c) They are unrelated in all crystal systems
- (d) [uvw] is parallel to the plane (hkl)
- (a) h + k
- (b) −(h + k) ✔
- (c) h − k
- (d) k + l
- (a) 4
- (b) 8 ✔ — (111), (1̄11), (11̄1), (111̄), (1̄1̄1), (1̄11̄), (11̄1̄), (1̄1̄1̄)
- (c) 6
- (d) 12
- (a) 0.2866 nm
- (b) 0.2027 nm ✔ — d = a/√(1²+1²+0²) = 0.2866/√2 = 0.2027 nm
- (c) 0.1655 nm
- (d) 0.4054 nm
- (a) 2
- (b) 3
- (c) 4 ✔ — (111), (1̄11), (11̄1), (111̄) are the 4 independent {111} planes, giving 4×3=12 FCC slip systems
- (d) 8
- (a) (110)
- (b) (111)
- (c) (001) ✔ — the c-axis intercept is 1, both a-axis intercepts are ∞; i = −(0+0) = 0
- (d) (100)
14. Key Takeaways
- PURPOSE: Miller indices provide a universal, compact notation system for identifying crystallographic planes (hkl) and directions [uvw] in any crystal, enabling unambiguous communication across all of materials science, physics, and engineering.
- 3-STEP PROCEDURE FOR PLANES: (1) Find intercepts on x, y, z axes as multiples of lattice parameters. (2) Take reciprocals of each intercept (1/∞ = 0). (3) Multiply by LCD to get smallest integers, enclose in parentheses (hkl).
- PARALLEL AXES: If a plane is parallel to an axis, its intercept is ∞ and its Miller index is 0. The (100) plane is parallel to both b and c axes.
- BAR NOTATION: A negative index (from a negative-side intercept) is written with a bar above the digit: 1̄ = "bar one". Example: (1̄10) is pronounced "bar-one, one, zero".
- ORIGIN RULE: If a plane passes through the origin, shift the origin to any other equivalent lattice point before applying the 3 steps.
- BRACKET NOTATION: (hkl) = specific plane; {hkl} = family of equivalent planes; [uvw] = specific direction; ⟨uvw⟩ = family of equivalent directions.
- CUBIC SPECIAL PROPERTY: In cubic systems only, direction [hkl] is perpendicular to plane (hkl). Not true in other crystal systems.
- MILLER-BRAVAIS (hkil): 4-index notation for hexagonal crystals. Always i = −(h + k). Reveals the 6-fold symmetry hidden by 3-index notation.
- KEY HCP PLANES: Basal (0001), prism (101̄0), pyramidal (101̄1) — each plays a different role in HCP deformation and slip.
- d-SPACING FOR CUBIC: d_hkl = a/√(h²+k²+l²). Combined with Bragg's Law, this links Miller indices directly to XRD peak positions.
- SELECTION RULES: BCC allows reflections only when (h+k+l) is even. FCC allows only when h,k,l are all odd or all even.
- ENGINEERING APPLICATIONS: Slip systems, semiconductor wafer orientation (Si is (100)), thin film texture, cleavage planes, XRD indexing — all described using Miller indices.
References
All references are in IEEE citation style. All sources are peer-reviewed journals, internationally recognised textbooks, or authoritative academic institutions.
- W. D. Callister Jr. and D. G. Rethwisch, Materials Science and Engineering: An Introduction, 10th ed. Hoboken, NJ, USA: John Wiley & Sons, 2018, ch. 3, pp. 60–75. [Wiley] — Primary textbook reference for the 3-step Miller index procedure, bar notation, families of planes, direction indices, and the cubic d-spacing formula.
- C. Kittel, Introduction to Solid State Physics, 8th ed. Hoboken, NJ, USA: John Wiley & Sons, 2005, ch. 1, pp. 9–18. [Wiley] — Reference for Miller indices in the context of reciprocal lattice, lattice planes, and the connection between (hkl) and diffraction theory.
- B. D. Cullity and S. R. Stock, Elements of X-Ray Diffraction, 3rd ed. Upper Saddle River, NJ, USA: Pearson Prentice Hall, 2001, ch. 2. — Definitive reference for Miller indices applied to XRD: d-spacing formulas, systematic absences for BCC and FCC, and the indexing of powder diffraction patterns.
- A. Kelly and K. M. Knowles, Crystallography and Crystal Defects, 2nd ed. Chichester, UK: John Wiley & Sons, 2012, ch. 1. [Wiley] — Reference for crystallographic directions, families of planes, Miller-Bravais notation for hexagonal systems, and the relationship between planes and slip systems.
- International Union of Crystallography (IUCr), "Teaching Pamphlets: Crystal Symmetry and Space Groups," IUCr Education Pamphlets, no. 9. Chester, UK: IUCr, 2002. [iucr.org — full text] — Authoritative reference for the formal definition of Miller indices, Miller-Bravais notation for hexagonal systems, and the relationship between crystal symmetry and plane equivalence.
- IUCr, "Powder Diffraction," IUCr Teaching Pamphlets. Chester, UK: IUCr. [iucr.org — X-ray powder diffraction] — Reference for BCC and FCC selection rules (systematic absences), d-spacing formulas for all crystal systems, and XRD peak indexing.
- N. W. Ashcroft and N. D. Mermin, Solid State Physics. Philadelphia, PA, USA: Holt, Rinehart and Winston, 1976, ch. 5. — Graduate reference for Miller indices in the context of reciprocal lattice vectors, Brillouin zones, and electronic structure calculations in cubic crystals.
- A. Jain et al., "Commentary: The Materials Project: A materials genome approach to accelerating materials innovation," APL Mater., vol. 1, no. 1, Art. no. 011002, 2013, doi: 10.1063/1.4812323. [Materials Project — Open Crystal Database] — Source for BCC iron lattice parameter (a = 0.2866 nm) and XRD peak positions used in the worked d-spacing example (Section 11, Example 6).
- R. Verma and S. K. Rout, "Frequency-dependent ferro–antiferro phase transition and internal bias field influenced piezoelectric response of donor and acceptor doped bismuth sodium titanate ceramics," J. Appl. Phys., vol. 126, no. 9, Art. no. 094103, Sep. 2019, doi: 10.1063/1.5111505. [AIP — DOI: 10.1063/1.5111505] — Author's peer-reviewed research employing XRD peak indexing using Miller indices to characterise structural phase transitions in BNT perovskite ceramics — directly relevant to the XRD application discussed in Section 9.
- R. Verma and S. K. Rout, "The Mystery of Dimensional Effects in Ferroelectricity," in Recent Advances in Multifunctional Perovskite Materials. London, UK: IntechOpen, 2022, doi: 10.5772/intechopen.104435. [IntechOpen — Open Access] — Author's chapter employing XRD and Miller index analysis to study crystallographic planes in perovskite thin films and their ferroelectric properties.
This tutorial is part of the Crystal Structure Hub Series at AdvanceMaterialsLab.com
Next in series: XRD & Bragg's Law — Crystal Structure Determination | Crystal Defects and Dislocations
© 2026 AdvanceMaterialsLab.com — Dr. Rolly Verma | All rights reserved
Dr. Rolly Verma
If you notice any inaccuracies or have constructive suggestions to improve the content, I warmly welcome your feedback. It helps maintain the quality and clarity of this educational resource. You can reach me at: advancematerialslab27@gmail.com
